Q1 Glycerol CCA ATP Acyl Call ADP NAD NADH+H+ Fully Acid Aceryl CoA Acetoacetyl CoA Acetyl CoA FAD L-Glycerol 3-phiphule ATP AMP+PP Dydroxyace phosphate Acyl CoA FADH H₂O + trans-Encyl CoA Acyl CoA (shared by 2 carbons) COA 요 H₂O + CA D-Glyceraldehyde 3-phosphate L-3-Hydroxyacyl CoA NAD H-NADH HS-COA COA 3-K CA 3-Hydroxy-3-methyl- glutaryl CoA Acetoacetate NACH+H NAD+ D-3-hydroxy- butyrate Apture
Q1 Glycerol CCA ATP Acyl Call ADP NAD NADH+H+ Fully Acid Aceryl CoA Acetoacetyl CoA Acetyl CoA FAD L-Glycerol 3-phiphule ATP AMP+PP Dydroxyace phosphate Acyl CoA FADH H₂O + trans-Encyl CoA Acyl CoA (shared by 2 carbons) COA 요 H₂O + CA D-Glyceraldehyde 3-phosphate L-3-Hydroxyacyl CoA NAD H-NADH HS-COA COA 3-K CA 3-Hydroxy-3-methyl- glutaryl CoA Acetoacetate NACH+H NAD+ D-3-hydroxy- butyrate Apture
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter24: Lipid Biosynthesis
Section: Chapter Questions
Problem 1P: Explaining the Stoichiometry of Fatty Acid Synthesis Carefully count and account for each of the...
Related questions
Question
I need help filling in the boxes
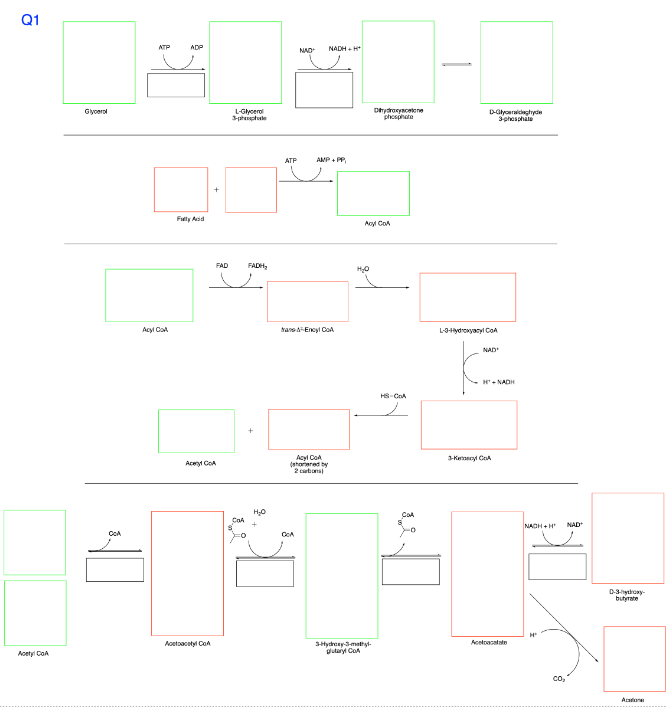
Transcribed Image Text:Q1
Glycerol
CCA
ATP
Acyl Call
ADP
NAD
NADH+H+
Fully Acid
Aceryl CoA
Acetoacetyl CoA
Acetyl CoA
FAD
L-Glycerol
3-phiphule
ATP
AMP+PP
Dydroxyace
phosphate
Acyl CoA
FADH
H₂O
+
trans-Encyl CoA
Acyl CoA
(shared by
2 carbons)
COA
요
H₂O
+
CA
D-Glyceraldehyde
3-phosphate
L-3-Hydroxyacyl CoA
NAD
H-NADH
HS-COA
COA
3-K CA
3-Hydroxy-3-methyl-
glutaryl CoA
Acetoacetate
NACH+H
NAD+
D-3-hydroxy-
butyrate
Apture
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax