Q2// To calculate the surface density of atoms on a particular plane in a crystal. Consider the body-centered cubic structure and the (110) plane shown in Figure 1.8a. Assume the atoms can be represented as hard spheres with the closest atoms touching each other. Assume the lattice constant is a = 5 Å. Figure 1.8b shows how the atoms are cut by the (110) plane. (а) (b)
Q2// To calculate the surface density of atoms on a particular plane in a crystal. Consider the body-centered cubic structure and the (110) plane shown in Figure 1.8a. Assume the atoms can be represented as hard spheres with the closest atoms touching each other. Assume the lattice constant is a = 5 Å. Figure 1.8b shows how the atoms are cut by the (110) plane. (а) (b)
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 107E
Related questions
Question
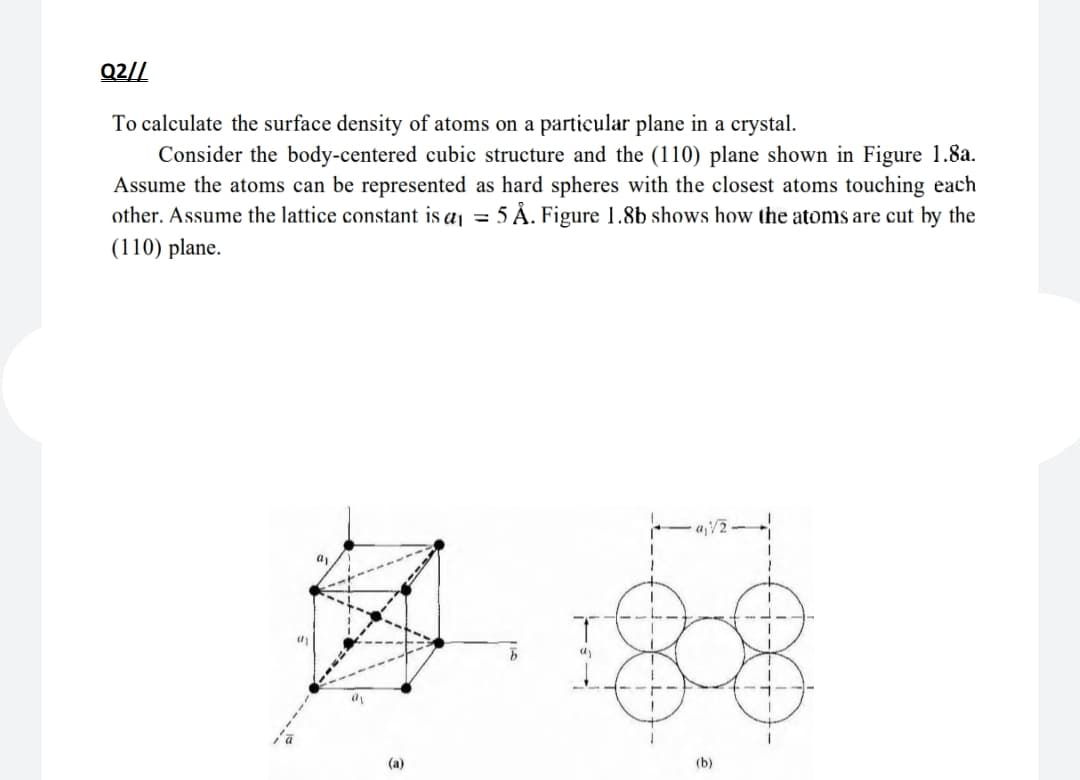
Transcribed Image Text:Q2//
To calculate the surface density of atoms on a particular plane in a crystal.
Consider the body-centered cubic structure and the (110) plane shown in Figure 1.8a.
Assume the atoms can be represented as hard spheres with the closest atoms touching each
other. Assume the lattice constant is a = 5 Å. Figure 1.8b shows how the atoms are cut by the
(110) plane.
a
(а)
(b)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning