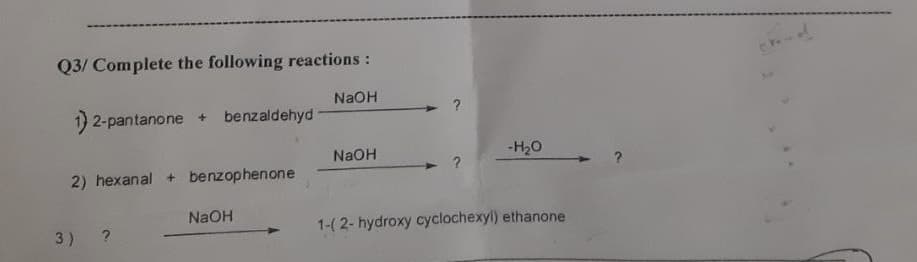
Q3/ Complete the following reactions: NaOH 1) 2-pantanone + benzaldehyd NaOH -H20 2) hexanal + benzophenone NaOH 1-(2- hydroxy cyclochexyl) ethanone 3)
Q: Problems: Please complete the questions in the space provided. Show all your work for full marks. 1.…
A:
Q: 1. H2SO4, heat 2. НCСІ, H-02 LOH d.
A:
Q: 1. For the following pair of molecules, indicate which molecule absorbs at the longest wavelength in…
A: According to woodward fisher rules A) homo annular diene = 253nm Ring residue = 4×5 = 20nm…
Q: The equilibrium constant for the dissociation of O, into atomic oxygen, 02 = 20 %3D at 3000 K is:…
A: The equilibrium reaction given is, => O2 ⇔ 2 O…
Q: 2. SAMPLE # 2 crucible, cover, unknown hydrate 18.1352g crucible and cover |5. \4० ४ sample 2.9944…
A: Mass of crucible, cover and unknown hydrate 18.1352 g Mass of crucible and cover 15.1408 g Mass…
Q: Which chemical test can distinguish 1-pentanol from 1-pentene? Explain
A: Alkene and alcohol both are hydrocarbon but Alkene contains double bond and alcohol contains -OH…
Q: Select all methods from the list below that can be used for the analytical determination of reducing…
A: Analytical determination of reducing agents
Q: What measurements or values will be needed to determine the molar mass of the unknown acid? mass of…
A:
Q: Percent Composition Practice Find the percent compositions of all of the elements in the following…
A:
Q: Where did the 0.0542 L from part e come from
A: At equivalence point, moles of acid is equals to moles of base.
Q: Reagents a. CeHsCHO b. NaOH, ethanol c. Pyrrolidine, cat. H* j. Br2, H* d. H2C=CHCN e. H3O* f. h.…
A: The reactant given is cyclopentanone and the product is,
Q: A student recorded the following end-point volumes in an iodometric titration using starch…
A: Titration error is -3.89 x 10-4 .
Q: At 25 °C, only 0.0810 mol of the generic salt AB2 is soluble in 1.00 L of water. What is the ?sp of…
A:
Q: How much heat is absorbed in the complete reaction of 3.00 grams of SiO, with excess carbon in the…
A:
Q: At a given temperature, the Keq for the reaction 2 HI(g) → H2(g) + I2(g) is 0.084 If the…
A:
Q: Find the rate law and the individual and overall reaction orders for the reaction H2 + 12 ---> 2HI…
A:
Q: B. Identify the IMFA present between molecules of each compound. Rank (from 1-3) the compounds in…
A:
Q: Green plants use light from the Sun to drive photosynthesis. Photosynthesis is a chemical reaction…
A: Weight of H2O = 5.6 gm
Q: 1. A student adds 25.27 mL of a 0.750 M Na2SO4 solution to another solution containing 2.50 grams of…
A: Given -> Volume of Na2SO4 = 25.27 ml Molarity of Na2SO4 = 0.750 M Weight of BaCl2 = 2.50 gm
Q: What volume would 50.0 g of propane, C3HS, occupy at 1.40 x 102°C under a pressure of 1.82 x 10…
A: Given, Mass of propane = 50.0 g Temperature (T) = 1.40 × 102°C = 140°C = (140 + 273.15) K = 413.15 K…
Q: Please predict major product
A:
Q: the treatment of mild cyanide poisoning
A:
Q: C. Solubility of Gases in Liquid Probable Observation Explanation Effect of Pressure |As you open…
A: Henry`s law is given by, solubility of a gas in a liquid is directly proportional to the partial…
Q: Consider these compounds: A. CPPO4 В. Mn(ОH)2 C. Caso3 D. BaF2 Complete the following statements by…
A: To compare the solubility of salts , we compare their Ksp values. Salt having higher Ksp would be…
Q: For the following reaction NO2(g) + SO2(g) NO(g) + SO3(g) K = 4.15 at a particular temperature. If…
A:
Q: Describe and map the reaction between Potassium dichromate (K2Cr2O7) and Sodium thiosulfate as a…
A: Potassium dichromate is the strong Oxidising agent which can easily oxidise different type of…
Q: For a certain acid pK, 2.34. Calculate the pH at which an aqueous solution of this acid would be…
A:
Q: ) Propose the possible substitution patterns for each of the derivatives based on the observed…
A: A) Based of splitting pattern, we can say that Derivative 1 1CH2 = 2CHX The hydrogen of carbon(1)…
Q: energy balance example discussed in class: A storage contains 0.2 m of liquid water at 75 C. The…
A: Solution -
Q: QUESTION 7 A kinetics experiment is performed to determine the activation energy of a reaction. The…
A: 7) The data given is,
Q: he statement "At constant temperature, the volume occupied by a definite mass of a gas is inversely…
A:
Q: erences] Draw a structural formula for the major organic product of the reaction shown. H3C CH3 C=C…
A:
Q: 17. An a helix or sheer is r exple A prinary semday peier arne wf ihes
A: 17. The structute of protein is divided into 4 levels. Primary, secondary, tertiary and quaternary…
Q: 4000 000 VENUE 1000
A: IR spectra deals results from the interaction of infrared radiation with matter by absorption. IR…
Q: 2. Which of the following correctly describes lipid properties? A. Lipids contain polar functional…
A:
Q: . Organometallic reagents are characterized by a. Polar bonds b. Carbon bonded to an…
A: ->organometallic compounds are compounds in which there is bond between carbon and…
Q: Above what Fe²+ concentration will Fe(OH), precipitate from a buffer solution that has a pH of 9.61?…
A: Given: pH of the solution = 9.61
Q: Which of the following statements is important when assessing a gravimetric analysis? the desired…
A: Precipitation of gravimetric method : The analyte is separated from a solution of the sample as a…
Q: Calculate the pH of the 1L buffer composed of 500 mL of 0.60 M acetic acid plus 500 mL of 0.60 M…
A:
Q: What happens to the volume of air confined in the syringe as the slotted masses are added? State the…
A: Boyle's law: Boyle's law is a gas law that states that a gas's pressure and volume are inversely…
Q: WORD SCRAMBLE Below are several scrambled words you have used in this Lesson. Unscramble the words…
A: Unscramble the given words and write the words according to them. In multiple questions we solve…
Q: :0-CH-CH3 C Hs- CHy-t-CH 3 Ha ÖCH,CH3 CHy
A:
Q: Given the equation Ag*(aq) + 2 NH, (aq) – [Ag(NH,),]*(aq) Kf = 2.00 × 107 determine the…
A: Given: Mass of AgCl to be dissolved = 529 mg = 0.529 g. Volume of solution = 100.0 mL = 0.100 L…
Q: According to this H NMR spectrum, identify the structure of the compound. the only additional…
A:
Q: Balancing: try both for practicel) Complete only ONE of the following questions for a reaction…
A: The reaction given is, => MnO4- + Fe2+ → Mn2+ + Fe3+
Q: Nitrogen and hydrogen react to form ammonia, like this: N2(g)+3H,(g) → 2 NH3(g) The reaction is…
A: Le Chatelier's principle: According to the Le Chatelier's principle when a change in concentration…
Q: During your lab titration of Q12*: At what pH has 50% of the reactant acid been titrated? Report…
A:
Q: What is the fraction of association (a) for each potassium propionate solution? The Ka of propanoic…
A:
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate…
A:
Q: 9. Use the standard free energy of formation data to determine the free energy change for the…
A: Balanced the equation and determine the free energy change for that reaction also identify the…
Step by step
Solved in 2 steps with 3 images

- Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.The base-promoted rearrangement of an a-haloketone to a carboxylic acid, known as the Favorskii rearrangement, is illustrated by the conversion of 2-chlorocyclohexanone to cyclopentanecarboxylic acid. Jo Han Sow La NaOH, THF O Nat H3O+ OH NaOH, THF The mechanism involves the following 5 steps: -ō cyclopropanone intermediate 1. Abstraction of a proton to form enolate anion 1; 2. Formation of a cyclopropanone intermediate 2 with expulsion of chloride ion; 3. Addition of hydroxide ion to form tetrahedral intermediate 3; 4. Collapse of the tetrahedral intermediate and breakage of the three-membered ring to form carbanion intermediate 4; 5. Proton transfer to form the rearranged carboxylic acid. For the following reaction, draw the reaction out on paper, and then draw the structure of cyclopropanone intermediate 2 in the window. 1. NaOH, THF 2. H3O* OHWhat would be a proposed mechanism to get from cyclohexanol to cis-2-methoxycyclohexanol? I know how to get from cyclohexanol to trans-2-methoxycyclohexanol would be to go through acid-catalyzed dehydration via H2SO4, then epoxide ring formation via mCPBA and then alcohol-catalyzed opening, but don't know how to do for the cis version, if possible.
- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?Minor product via E1 for 2-butanol and HCl?Thank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 Isobutalene
- Provide the mechansim using curved arrows of the reaction of p-t-butyl phenol treated with acetic anhydride in AlCl3. Include resonance stabilized intermediates and if more than one product is formed, label them as major, minor, etc. thank you for the helpWhat steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Predict the products of reaction of pent-1-yne with the following reagents. cold, dilute KMnO4
- Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:The reaction of 2-ethyl-1-pentene with Br2, with H2 + Pd/C, or with R2BH/THF followed by aqueous HO- + H2O2 leads to a racemic mixture. Explain why a racemic mixture is obtained in each case.Please describe the difference in the outcome of treating a terminal alkyne with HgSO4, H2SO4, H2O and with 9-BBN, followed by H2O2 and NaOH.

