C. Solubility of Gases in Liquid Probable Observation Explanation Effect of Pressure |As you open the bottle of a soft drink. Probable Observation Explanation Effect of Temperature As the soft drink is heated At what condition should gases be dissolved in liquid? Give at least 3 human activities that would increase the bodies of 1. water temperature (thermal pollution) that will lead to the decrease of the solubility of oxygen and carbon dioxide gas in bodies of water, which are needed by aquatic plants and animals respectively. 3. 2.
C. Solubility of Gases in Liquid Probable Observation Explanation Effect of Pressure |As you open the bottle of a soft drink. Probable Observation Explanation Effect of Temperature As the soft drink is heated At what condition should gases be dissolved in liquid? Give at least 3 human activities that would increase the bodies of 1. water temperature (thermal pollution) that will lead to the decrease of the solubility of oxygen and carbon dioxide gas in bodies of water, which are needed by aquatic plants and animals respectively. 3. 2.
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter10: Properties Of Solutions
Section: Chapter Questions
Problem 7RQ
Related questions
Question
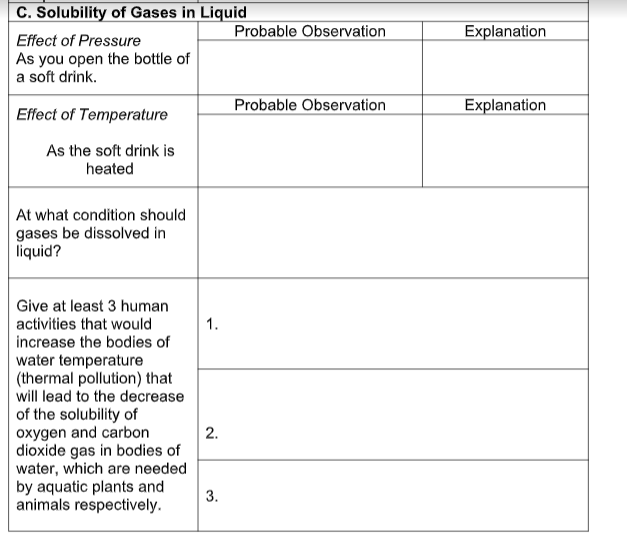
Transcribed Image Text:C. Solubility of Gases in Liquid
Probable Observation
Explanation
Effect of Pressure
As you open the bottle of
a soft drink.
Probable Observation
Explanation
Effect of Temperature
As the soft drink is
heated
At what condition should
gases be dissolved in
liquid?
Give at least 3 human
activities that would
increase the bodies of
1.
water temperature
(thermal pollution) that
will lead to the decrease
of the solubility of
oxygen and carbon
dioxide gas in bodies of
water, which are needed
by aquatic plants and
animals respectively.
2.
3.

Transcribed Image Text:1. Effect of Pressure
Considering that you
a. Open a bottle of soft drink. What do you think will be produced? Describe the
sound that is produced.
b. Note if any bubbles are formed.
c. Record your observations in the data sheet.
2. Effect of Temperature
Take for instance you
a. Transfer 50.0 mL of the soft drink in a 100-mL beaker and you
b. Heat it to about 50°C.
c. Observed if bubbles are formed
c. Record your observation.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning