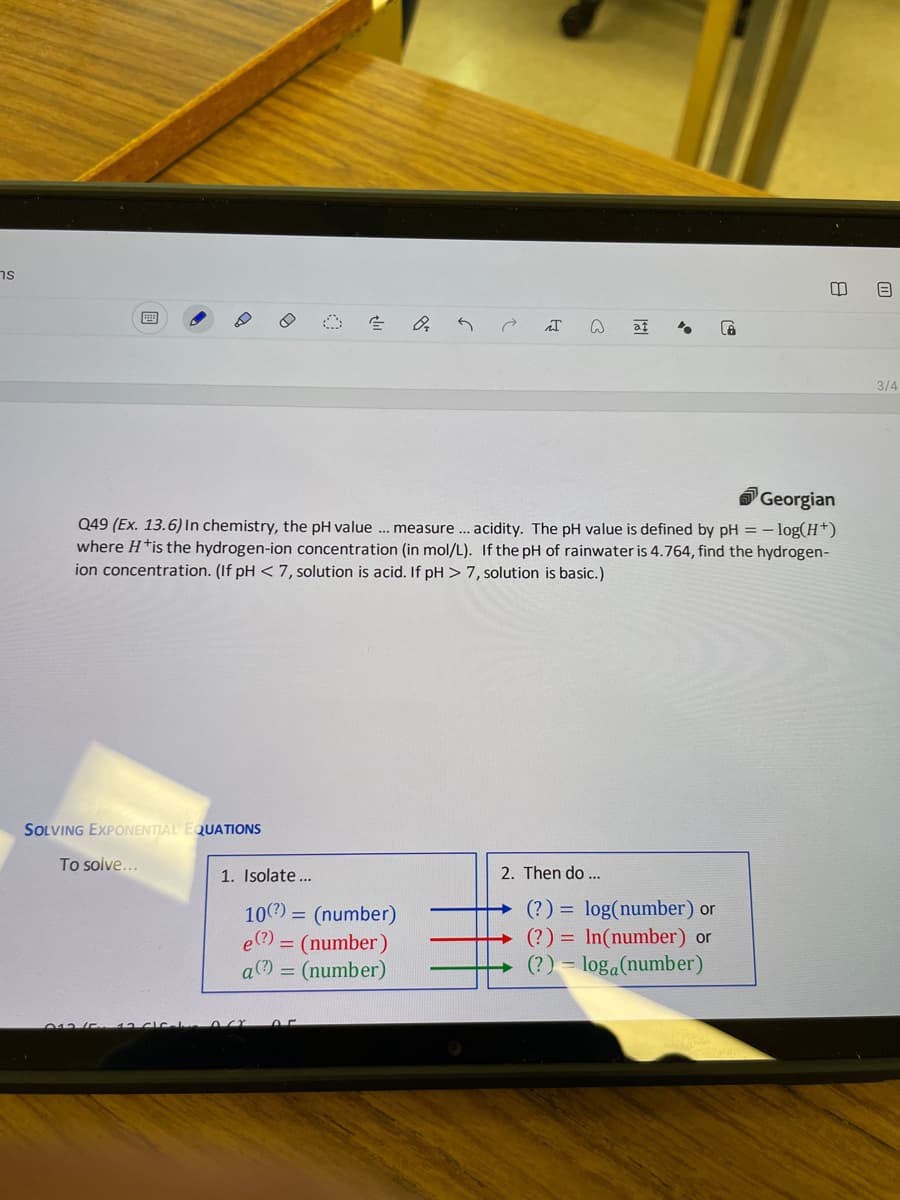
Q49 (Ex. 13.6) In chemistry, the pH value . measure.. acidity. The pH value is defined by pH where Htis the hydrogen-ion concentration (in mol/L). If the pH of rainwater is 4.764, find the hydrogen- ion concentration. (If pH < 7, solution is acid. If pH > 7, solution is basic.) - log(H*)
Q49 (Ex. 13.6) In chemistry, the pH value . measure.. acidity. The pH value is defined by pH where Htis the hydrogen-ion concentration (in mol/L). If the pH of rainwater is 4.764, find the hydrogen- ion concentration. (If pH < 7, solution is acid. If pH > 7, solution is basic.) - log(H*)
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.16QAP
Related questions
Question
Transcribed Image Text:ns
画
ది
3/4
Georgian
Q49 (Ex. 13.6) In chemistry, the pH value .. measure.. acidity. The pH value is defined by pH = – log(H*)
where H*is the hydrogen-ion concentration (in mol/L). If the pH of rainwater is 4.764, find the hydrogen-
ion concentration. (If pH < 7, solution is acid. If pH > 7, solution is basic.)
SOLVING EXPONENTIAL EQUATIONS
To solve...
1. Isolate...
2. Then do ...
(?) = log(number) or
(?) = In(number) or
(?) = loga(number)
10?) = (number)
e(?)
= (number)
a(?) = (number)
012/5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT