Question 1 1 pts Mrs. Vereen was given a huge gemstone that looked like a diamond. The task was to determine if it was real or a fake (made of cubic zirconium). The enormous stone weighed 2 g. When placed in a graduated cylinder of water, the water level went up by .60 mL. The density of a real diamond is 3.0-3.5 g/mL. Determine the density of the stone Mrs. Vereen given.
Question 1 1 pts Mrs. Vereen was given a huge gemstone that looked like a diamond. The task was to determine if it was real or a fake (made of cubic zirconium). The enormous stone weighed 2 g. When placed in a graduated cylinder of water, the water level went up by .60 mL. The density of a real diamond is 3.0-3.5 g/mL. Determine the density of the stone Mrs. Vereen given.
College Physics
1st Edition
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:Paul Peter Urone, Roger Hinrichs
Chapter12: Fluid Dynamics And Its Biological And Medical Applications
Section: Chapter Questions
Problem 9PE: (a) Estimate the time it would take to fill a private swimming pool with a capacity of 80,000 L...
Related questions
Question
Please help

Transcribed Image Text:Question 1
1 pts
Mrs. Vereen was given a huge gemstone that looked like a diamond.
The task was to determine if it was real or a fake (made of cubic
zirconium).
The enormous stone weighed 2 g.
When placed in a graduated cylinder of water, the water level
went up by .60 mL.
The density of a real diamond is 3.0-3.5 g/mL.
Determine the density of the stone Mrs. Vereen given.
Transcribed Image Text:ing. All. Li.
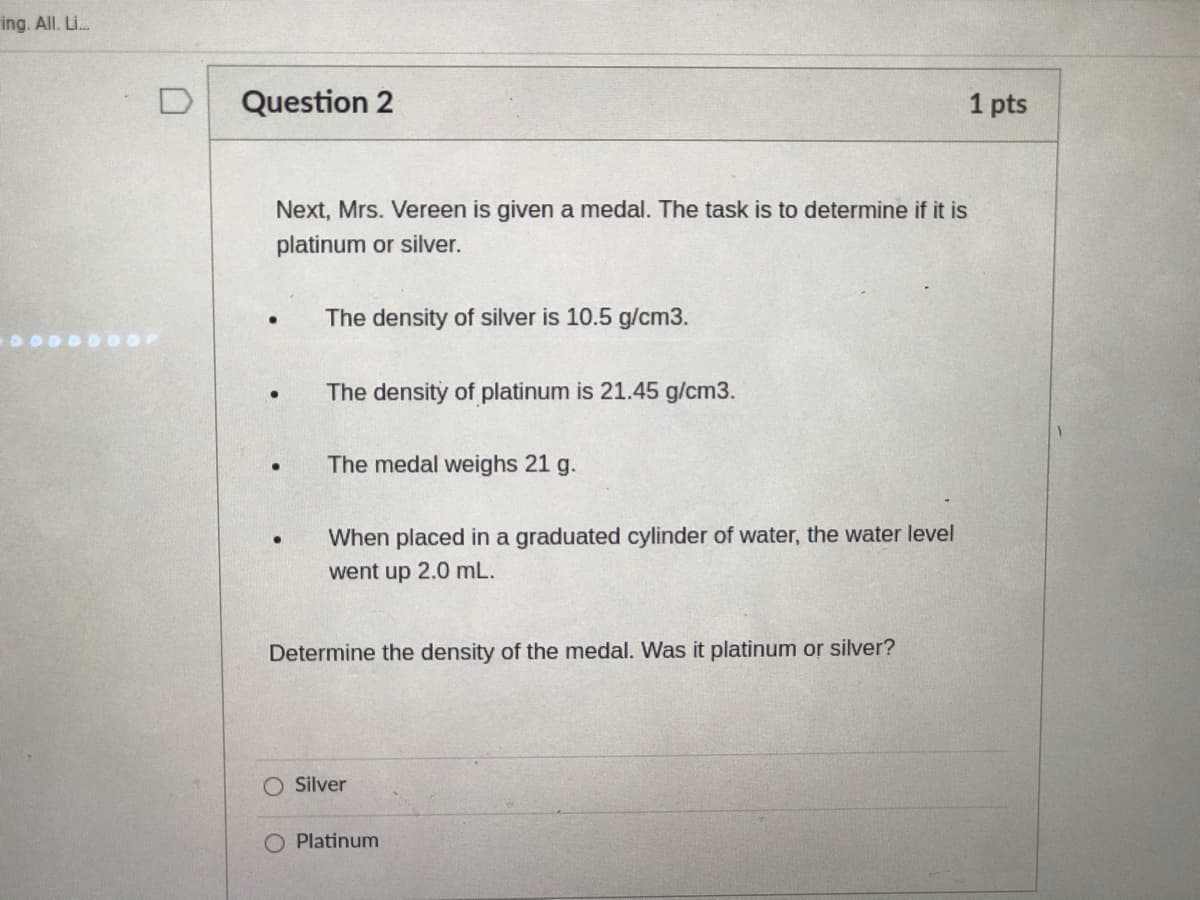
Question 2
1 pts
Next, Mrs. Vereen is given a medal. The task is to determine if it is
platinum or silver.
The density of silver is 10.5 g/cm3.
The density of platinum is 21.45 g/cm3.
The medal weighs 21 g.
When placed in a graduated cylinder of water, the water level
went up 2.0 mL.
Determine the density of the medal. Was it platinum or silver?
Silver
Platinum
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781305952300
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning