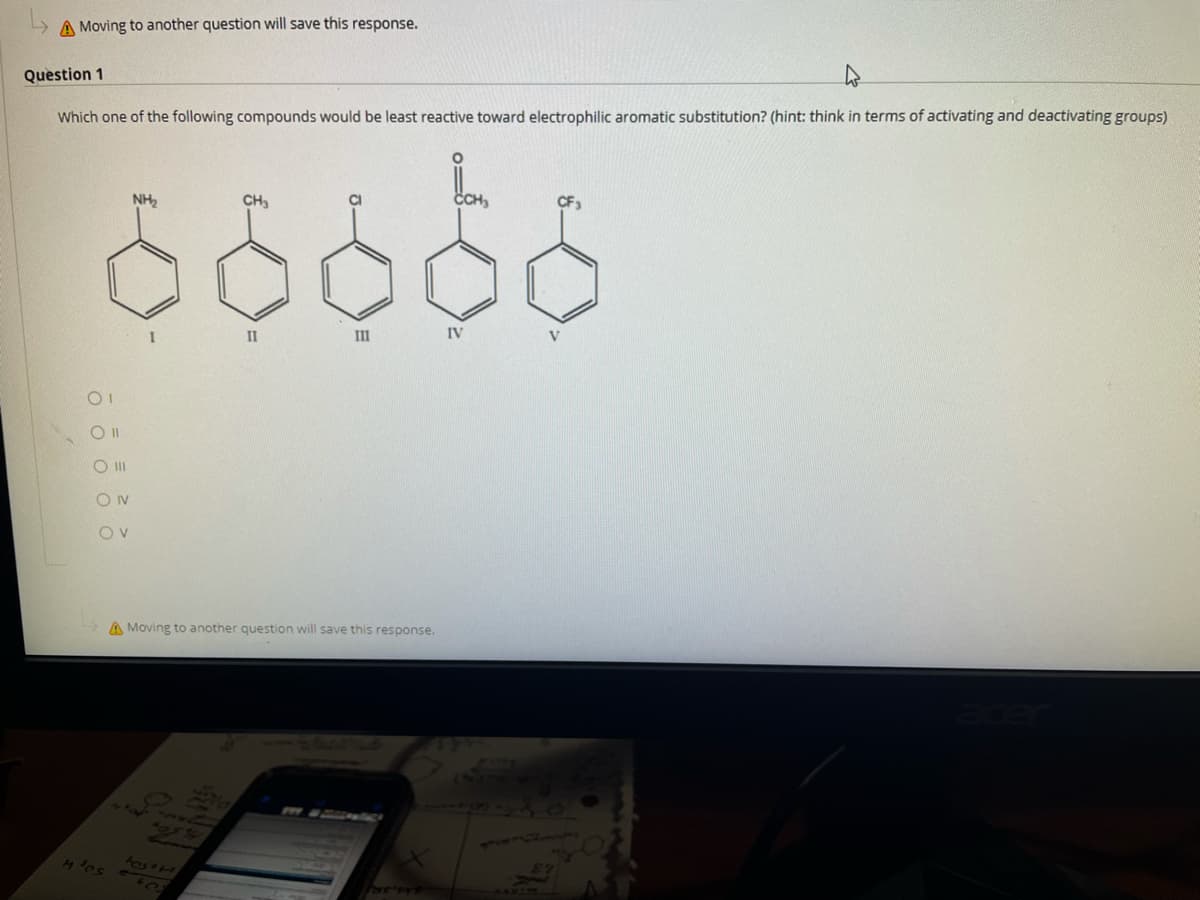
Question 1 Which one of the following compounds would be least reactive toward electrophilic aromatic substitution? (hint: think in terms of activating and deactivating groups) NH, CH, CH, II IV V O II 0 0 0 0 0
Question 1 Which one of the following compounds would be least reactive toward electrophilic aromatic substitution? (hint: think in terms of activating and deactivating groups) NH, CH, CH, II IV V O II 0 0 0 0 0
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter18: Aromaticity
Section: Chapter Questions
Problem 29CTQ
Related questions
Question
Transcribed Image Text:L> A Moving to another question will save this response.
Question 1
Which one of the following compounds would be least reactive toward electrophilic aromatic substitution? (hint: think in terms of activating and deactivating groups)
NH2
CH3
CI
CH,
CF3
II
II
IV
V
A Moving to another question will save this response.
4034
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning