QUESTION 10 A reaction A→B has a second order rate law with k= 8.75×10-2Lmol-1s-1 at 30°C. Calculate the time (s) required for the concentration of A to change from 0.360 moll-1 to 0.025molL-1. Please round to nearest integer. For example, 10.7 is 10. 3600 QUESTION 11 Regarding the reaction described above, if the temperature dependence of rate constant obeys Arrhenius equation and the activation energy is 60.5kJ mol-1, calculate the rate constant (s) at 50°C. Please round to two decimal places.
QUESTION 10 A reaction A→B has a second order rate law with k= 8.75×10-2Lmol-1s-1 at 30°C. Calculate the time (s) required for the concentration of A to change from 0.360 moll-1 to 0.025molL-1. Please round to nearest integer. For example, 10.7 is 10. 3600 QUESTION 11 Regarding the reaction described above, if the temperature dependence of rate constant obeys Arrhenius equation and the activation energy is 60.5kJ mol-1, calculate the rate constant (s) at 50°C. Please round to two decimal places.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter11: Chemical Kinetics: Rates Of Reactions
Section: Chapter Questions
Problem 11.ACP: (Section 11-5) A rule of thumb is that for a typical reaction, if concentrations are unchanged, a...
Related questions
Question
can u check is 1st answer is correct and help me solve 2nd answer thanks
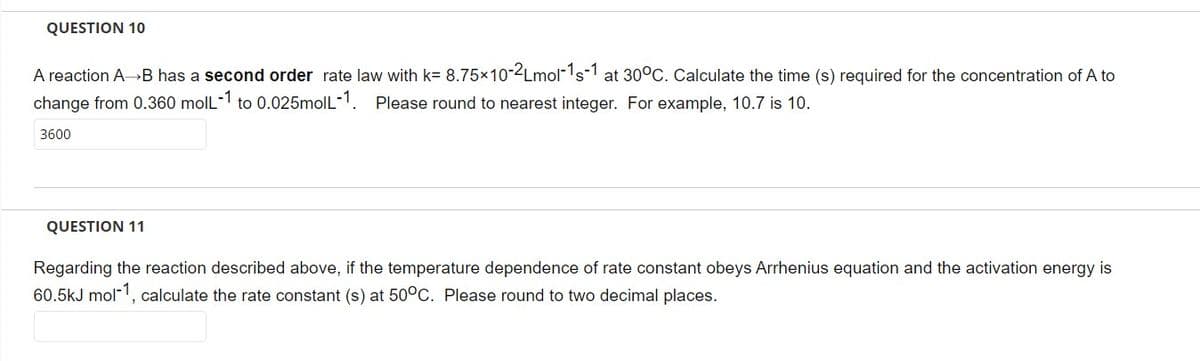
Transcribed Image Text:QUESTION 10
A reaction A→B has a second order rate law with k= 8.75×10-2Lmol-1s-1 at 30°C. Calculate the time (s) required for the concentration of A to
change from 0.360 moll-1 to 0.025moll-1. Please round to nearest integer. For example, 10.7 is 10.
3600
QUESTION 11
Regarding the reaction described above, if the temperature dependence of rate constant obeys Arrhenius equation and the activation energy is
60.5kJ mol-1, calculate the rate constant (s) at 50°C. Please round to two decimal places.
Expert Solution
Step 1
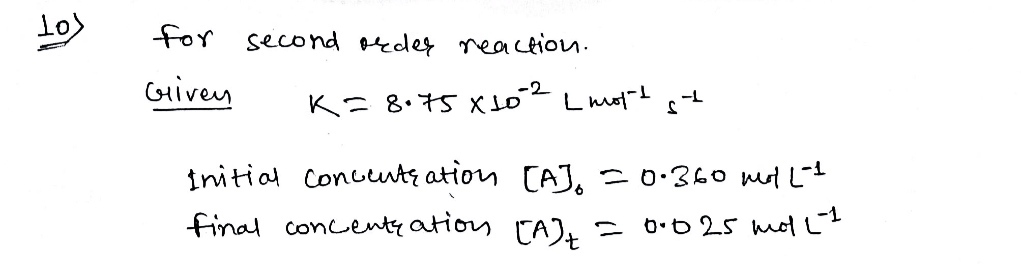
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning