QUESTION 10 If SL of wet O2 is collected over water at 25°C and a barometric pressure of 800 Torr, how many moles of O2 are present? O 14 0 14 O 14 O 140 QUESTION 11 A mixture of hydrogen (2 01 g) and chlorine (17.73 g) in a container at 300 K has a total gas pressure of 98.8 kPa What is the partial pressure of hydrogen in the mixture? O 794 kPa O 794 kPa O704 IPa O 7,940 kPa QUESTION 12 What is a polymor that forms by simple linking of monomers without the co-generation of other products O Addition Polymers O Polymers O Dehydration Polymers O Condensation Polymers
QUESTION 10 If SL of wet O2 is collected over water at 25°C and a barometric pressure of 800 Torr, how many moles of O2 are present? O 14 0 14 O 14 O 140 QUESTION 11 A mixture of hydrogen (2 01 g) and chlorine (17.73 g) in a container at 300 K has a total gas pressure of 98.8 kPa What is the partial pressure of hydrogen in the mixture? O 794 kPa O 794 kPa O704 IPa O 7,940 kPa QUESTION 12 What is a polymor that forms by simple linking of monomers without the co-generation of other products O Addition Polymers O Polymers O Dehydration Polymers O Condensation Polymers
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter5: Gases, Liquids, And Solids
Section: Chapter Questions
Problem 5.25P: 5-25 A gas in a bulb as in Figure 5-3 registers a pressure of 833 mm Hg in the manometer in which...
Related questions
Question
Give handwritten answer. ASAP
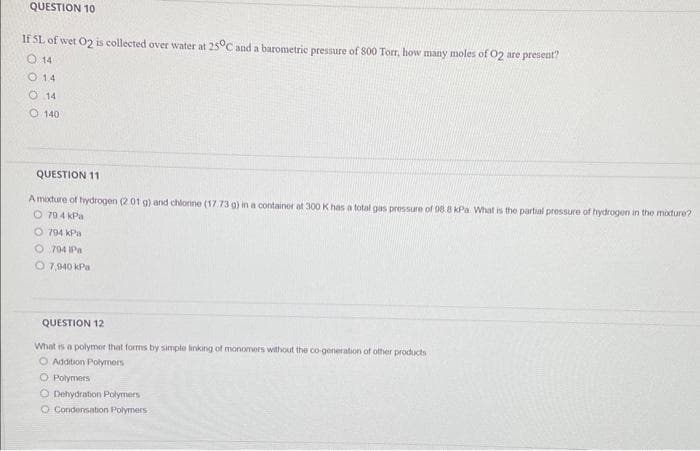
Transcribed Image Text:QUESTION 10
If SL of wet O2 is collected over water at 25°C and a barometric pressure of 800 Torr, how many moles of O2 are present?
O 14
0 14
O 14
O 140
QUESTION 11
A mixture of hydrogen (2 01 g) and chlorine (17.73 g) in a container at 300 K has a total gas pressure of 98.8 kPa What is the partial pressure of hydrogen in the mixture?
O 794 kPa
O 794 kPa
O704 IPa
O 7,940 kPa
QUESTION 12
What is a polymor that forms by simple linking of monomers without the co-generation of other products
O Addition Polymers
O Polymers
O Dehydration Polymers
O Condensation Polymers
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co