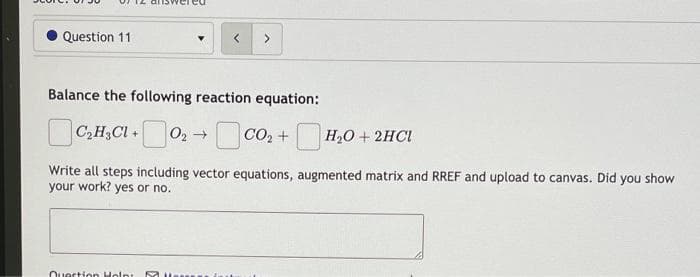
Question 11 Balance the following reaction equation: C,H3CI + C,H3CI+02 CO2 + H2O+ 2HCI Write all steps including vector equations, augmented matrix and RREF and unload t
Q: The literature value for the Ksp of Ca(OH)2 is 5.5e-6 at 25 oC. How does this compare with the value…
A: The Ksp value of calcium hydroxide is 5.5×10-6 and given that value obtained from the experiment is…
Q: A tank with 43.2 kg of water is leaking at a rate of 0.0135 kg/s. How many hours will it take until…
A: We have to calculate time to empty the tank.
Q: A unique plant was discovered from the hot springs of Maquinit, Coron, Palawan. To study the…
A: Answer is attached below
Q: L000 kg of calcium phosphate is made to react with 10% excess silica, SiOz and 90% excess carbon to…
A: The law of conservation of mass states that “ mass in an isolated system is neither created nor…
Q: To calculate the unknown concentration of a chemical in a solid sample, 4 different calibration…
A: To determine the unknown concentration, it's great to have a linear curve so that the property (such…
Q: Based on the chemical reactions provided, which of the following is equivalent to KMg-EDTA? [Mg2*]…
A: KCa-EDTA , KMg-Ind , KMg-EDTA are the equilibrium constants for complex formation. The overall…
Q: Organic carbon in seawater can be measured by oxidation to CO2 with K2S2O8, followed by gravimetric…
A:
Q: How many mL of 1:5000 (w/v) solution of phenylmercuric acetate can be made from 125 mL of 0.2%…
A:
Q: A tank has a total volume of 2.1 ft^3 It is pumped out and filled with 20 lbm of CO2 and 10 lbm of…
A: Since 1 Kg = 2.2 lbm And 1 Kg = 1000 g Hence 2.2 lbm = 1000 g => 1 lbm = 454.55 g Hence mass in…
Q: Which of the following are equivalent to 2,500 ppm Cu2+? MW: Cu (63.55) 2.5 ppb Cu2+ 2,500,000 ppb…
A: Given: Concentration of Cu2+ = 2500 ppm Molecular weight of Cu = 63.55 g/mol
Q: Principle: Direct permanganometry The percentage purity of hydrogen peroxide set by the USP is 2.5 –…
A: Given: Volume of H2O2 = 2.0 mL density of = 1.45 g/mL Concentration of KMnO4 = 0.1 N
Q: Solve the problem attached
A: We have to find the Vmax, Km and Kcat of given plot
Q: Use only the first decimal points (X.X) for atomic masses. R = 8.314 L*kPa/mole*K. In a reactor at…
A: The given reaction is: The limiting reactant can be found out by the following steps: 1. The molar…
Q: Condition Column I Comparison Column II For the reaction: 2SO3(g) = Кр [ Select ] Kc 2SO2(g) + O2(g)…
A:
Q: session.masteringchemistry.com/myct/itemView?assignmentProblemiD=144082727 CHE154-H Gen Chem II…
A: 1) Write the balanced chemical equation. 2) Where, N = normality
Q: A unique plant was discovered from the hot springs of Maquinit, Coron, Palawan. To study the…
A: The graph of calibration is given below.
Q: MULTIPLE CHOICE Question 5 Which statement best describes the major functionality difference between…
A:
Q: A 50.00 (±0.03) mL portion of an HCl solution required 29.71(±0.03) mL of 0.01963(±0.0030) M Ba(OH)2…
A: Given, MHCl=29.71 ml×0.01963 mmol Ba(OH)2ml×2mmol HClmmol Ba(OH)250.00 ml
Q: Given the following unbalanced response: NO2-+MnO4-+H+ NO3-+Mn2++H2O What is the number of…
A: In a balanced reaction, there must be an equal number of atoms of all elements on both sides of the…
Q: 47.6 mL aliquot from a 0.5 L solution that contains 0.45g of MnSO4 (MW is 151 g/mol) required 41.9…
A:
Q: Concentration (mg/ml) Time (min) 8.5 0.5 6.8 1 5.4 1.5 4.0 2 3.2 2.5 2.5 3 What will be the slope of…
A: Concept used: Slope = (Y2 - Y1)/ (X2 - X1) y = mx + c
Q: Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer)…
A: Given data 2500 ppm equal to
Q: How do I solve this equation, including the symbols for the states?
A: The standard formation reaction has a mole of compound as its only product and for reactants has…
Q: v Show Time Remaining ▼ A 0.45 M KBr solution OA contains 0.45 milliliters of KBr in 1 liter of the…
A: Molarity is defined as the number of moles of solute in 1 Liter of the solution. Molarity of…
Q: You need to make 500.0mL of 3.50 M NaOH from a stock solution of 8.25M NaOH. How many mL of 8.25M…
A: Molarity of NaOH (stock Solution ) = M1 = 8.25 MVolume of stock solution (V1) = To be detemined…
Q: AST ELIN nasal spray contains 0.1% azelastine hydrochloride and 125 mcg/mL of benzalkonium chloride…
A: The nasal spray contains 0.1% azelastine hydrochloride, which means 100 mL of spray contains 0.1 g…
Q: What would be the effect (+, - or 0) on the calculated Rexp for each of the following? Please…
A: Answers are explained below. The effect on calculated Rexp for each case is as follows:
Q: Consider a pond with three types of fish. 1) N1= 100, M1-1 lb 2) N2-30, M2-3 lb 3) N3-10, M3-7 lb…
A: The solution is given below. Q1. 3.3077 lb Q2. 5.0465 lb Q3. 1.8571
Q: ICan Do It Exercise 1: In the below setup, iron is added to acid in a flask fitted with empty…
A: The basis for above question is that iron is more reactive than hydrogen so it replaces hydrogen…
Q: A 10 ml of concentrated acetic acid was added in a sufficient quantity of water to make 350 ml of…
A:
Q: 101 Chem101 O 8 https://app.101edu.co Question 4 of 9 A 2.00 L graduated cylinder contains 1.40 L of…
A: Density of solution can be defined as mass of solution per unit volume of that solution. Litre and…
Q: 4.00 (±0.01) mL4.00 (±0.01) mL Class A transfer pipet is used to transfer 4.00 mL4.00 mL of a 0.300…
A: While diluting a solution, the dilution equation used is: M1V1 = M2V2
Q: Mixture Flow Problem A tank contains 2000 L of brine. Brine containing 5N/L of salt flows in at…
A:
Q: Question 21 Calculate the pressure difference (in Pa) that must be generated by a vertical drinking…
A:
Q: 25L bag was filled with air, a 10uL sample was then injected into the bag to make 50ppmv vapor…
A:
Q: QUESTION 8 Were the sample prep and the standard prep supplemented with the same amount of internal…
A: An internal standard in analytical chemistry is the substance similar to the unknown sample that is…
Q: Limiting reagent Calculated mass of excess reagent remaining in the mixture after reaction, g…
A:
Q: Can you work our KSP for me? thanks Liquid Amount - How many grams of each of the following…
A: Here given that the The solubility is 0.123 g/100 mL So substance dissolve in 4.70×102 mL of cold…
Q: QUESTION 13 The following data was collected during an OBLA test. What is the OBLA Workload in Watts…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: The test tube used in the determination of the ΔHrxn for nitric acid was still wet with water. […
A: 1) The test tube used in the determination of the ΔHrxn for nitric acid was still wet with water. […
Q: PHA 0175 Lab 4.1 Compounding Ointment Calculation Pharmaceutical Compounding Master Formula Question…
A: A numerical problem based on dimensional analysis that is to be accomplished.
Q: 12.10-3. Countercurrent Multistage Washing of Ore. A treated ore containing inert solid gangue and…
A: Soln:- No, of stages :-X0=120010,000 = 0.12R0 = 10,000 kg/ hr.V1=89.2 = 0.0869By center current…
Q: The test tube used in the determination of the AHrxn for nitric acid was still wet with water. […
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 5. Given: Cr| Cr3+ || Pb2+ | Pb ecell when pPb = 2 + ecell when pPb = 45 At the same temperature […
A: Given Cell Cr/Cr3+ //Pb2+/Pb
Q: Organic carbon in seawater can be measured by oxidation to CO2 with K2S2O8, followed by gravimetric…
A: Molar mass is defined as an average mass of atoms present in the chemical formula. It is the sum of…
Q: Question 12: Split injection in GC entails that some of the eluate from the column is split off and…
A: Gas chromatography (GC) is an analytical technique for separating chemical components of a sample…
Q: Standardization of EDTA Solution Answer the concentrations in 3 significant figure. You can use…
A: First, we have to calculate the molarity of the stock and working standard CaCO3 solution.
Q: A 1.000-g sample of unknown gave 2.500 g of bis(dimethylglyoximate)nickel(II) when analyzed by…
A: Solution : Weight percent is the amount of solute expressed in gram in 100 g of solution. It…
Q: What is the capital cost of a plant that produces 250,000 tons per year of ethanol (fuel grade)…
A: Ans) $ 149.89 M
Q: Kavh Search... ge.com/coursehimicourseld 154322748HeplD-2b3e48e6520860bf d5591538a4a5a27b#10001…
A: Any diatomic gas contains 22.4L of volume at STP.
Please provide me answer with vector equation, augmented matrix and RREF.
I will really upvote.
Thanks
Step by step
Solved in 3 steps

- Please show steps and states (aq),(s).. etc thank youPls help me with the following and make sure its 100% its important asap, thank you smChemistry pls help, I dont understand how this software wants me to enter the answer. its asking lfr the starting amount but its not taking it in the way Im putting it
- Calculate Trial I, Trial II, Trial III Vol.Ag(NO3)Added.Here's the link https://www.youtube.com/watch?v=wY3ey4E-FRAlt ls desired t0 c0ncentrate a 6% KNO3 s0luti0n in water t0 20% KNO3 s0luti0n. 7000 kg of pr0duct liqu0r is t0 be needed/hr. H0w much s0luti0n must be charged? H0w much water is evap0rated?
- Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer) MW: Cu (63.55) a) 2.5 ppb Cu2+ b) 2,500,000 ppb Cu c) 2.5 ppt Cu2+ d) 39.34 mM e) 0.03934 M f) 0.07868 N (in precipitation reaction) g) 0.07868 N (in redox into Cu+)Balance each of the chemical equations shown below. Be sure to include values of 1 for any species that only have 1 equivalent involved in the reaction (i.e. empty inputs DO NOT correspond to the value "1"). Use integers for ALL coefficients.Please answer atleast 4 sub-parts. Thank you.
- Which of the following is a spontaneous reaction.? a. Rxn with ΔH =- 10Kj/mol ΔS= -5J/mol T= 300K b. NaCl +H20 -> NaOH + HCl 25C c. H20(l) -> H2O(s) Temp: 25C d. Dissolution of 100g of solid sugar in 100 mL ice tea. Consider following reaction: HgO (s) -> Hg(l) + ½ O2 (g) Delta H = +90.7 kj/mol. What quantity of heat in kj/mol is required to produce one mole HgO? Write your answer without units. Given the following data 2ClF(g) + O2(g) --> Cl2O(g) + F2O (g) Delta H= 167.4 kJ I 2ClF3(g) + 2O2(g) --> Cl2O(g) + 3F2 O (g) Delta H= 341.4 kJ II 2 F2(g) + O2(g) ---> 2F2O (g) Delta H= -43.4 kJ III Calculate the delta H in kJ for below reaction: ClF(g) + F2(g) ---> ClF3(g)Jansen Gas creates three types of aviation gasoline(avgas), labeled A, B, and C. It does this by blendingfour feedstocks: Alkylate; Catalytic CrackedGasoline; Straight Run Gasoline; and Isopentane.Jansen’s production manager, Dave Wagner, hascompiled the data on feedstocks and gas types inTables 4.6 and 4.7. Table 4.6 lists the availabilitiesand values of the feedstocks, as well as their keychemical properties, Reid vapor pressure, and octanerating. Table 4.7 lists the gallons required, theprices, and chemical requirements of the three gastypes. Table 4.6 Data on Feedstocks Feedstock Alkylate CCG SRG Isopentane Gallons available (1000s) 140 130 140 110 Value per gallon $4.50 $2.50 $2.25 $2.35 Reid vapor pressure 5 8 4 20 Octane (low TEL) 98 87 83 101 Octane (high TEL) 107 93 89 108 Table 4.7 Data on Gasoline Gasoline A B C Gallons required (1000s) 120 130 120 Price per gallon $3.00 $3.50 $4.00 Max Reid pressure 7 7 7 Min octane 90 97 100 TEL level Low High High Note that each feedstock…Sent this question twice and Its gotten rejected, because it says something about honour code. this question is a completion question and its ungraded. Its only for completion. Please kindly work this solution.

