Q: 1. 2. OH CH3 H₂C+ CH3 1. 2. + + Na HCO3 a Proton transfer = H₂O CH3 H₂CH₂ CH3 Na + CO₂ H₂O d =…
A: We will need to identify the mechanism of reaction for forming the products by which we can tell…
Q: Table 1. Mass calculations for copper electrode. Mass of copper (initial) (g) Mass of copper (final)…
A: The objective of the first question is to calculate the partial pressure of the hydrogen gas that…
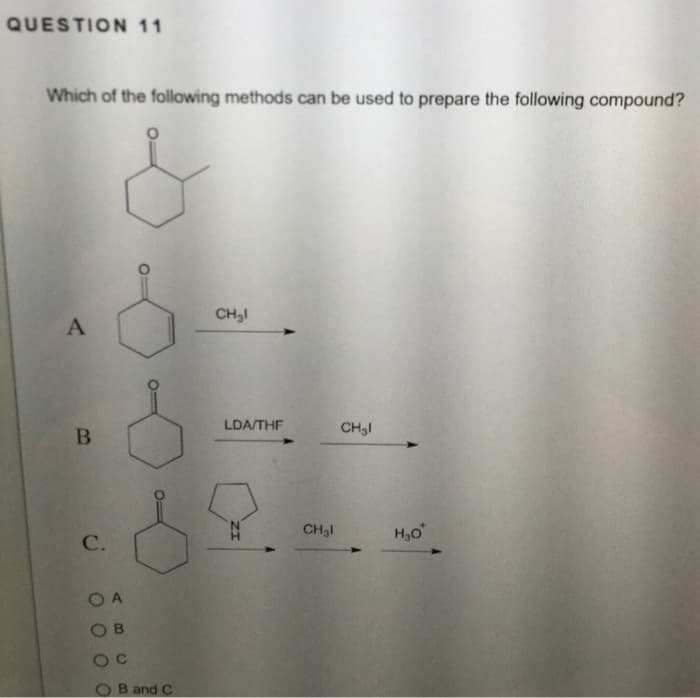
Q: be: The ester shown below can be formed by a Dieckmann cyclization. The starting material would Joa…
A: The objective of the question is to choose the starting material for the formation of the given…
Q: The answer is E. Please explain. Thanks.
A: The objective of this question is to determine the mass of silver that will be produced in the…
Q: Complete and balance the following half-reaction in acidic solution. Be sure to include the proper…
A: In an acidic solution, every excess oxygen atom is balanced by adding H2O molecules on the side…
Q: Water is critical to understand for biophysical processes. a. Draw the chemical structure of water…
A: The objective of this question is to explain the pH of water and its bond angle. The number of water…
Q: 2. The complex [Mo(CNH)8]** is an eight-coordinate complex discussed in Angew. Chem. Int. Ed. 2020,…
A: The aim of this question is to explain the complex [Mo(CNH) 8 ] 4 * contains isocyanate ligands, and…
Q: For the series of reactions below, what is the overall reaction and the value of ∆Go’(kJ/mol) for…
A: The objective of this question is to show the determine the overall reaction and the standard Gibbs…
Q: If the starting molecule has the following stereochemistry, what will be the stereochemistry of the…
A: Halide molecules and alkyl halide in the presence of heat or light undergo free radical reaction. A…
Q: 3. Using only acetaldehydes and acetones as your carbon sources, propose a mechanism to synthesize…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Give the systematic name for the following coordination compound or complex ion: CI- Co F
A: The main aim of this question is to explain the systemic name of given coordination complex. It…
Q: Groundwater containing 0.0004 M of DDT (molecular weight =354.5 g/mol) is added to a silt loam soil…
A:
Q: What mutant of Dihydroorotase affected the most it's turnover rate? Estimate its impact on the…
A: The objective of this question is to show the mutants is one that has a significant effect on…
Q: 7) Uranium-238 decays by alpha emission to form a new nuclide. Identify the new nuclide. A)…
A: The main aim of this question is to show the Uranium-238 decays by alpha emission to form a new…
Q: When the Pb²+ concentration is 3.44×104 M, the observed cell potential at 298K for an…
A: The objective of the question is to find the concentration of Mg²⁺ in an electrochemical cell given…
Q: Ground state 242 Cm undergoes alpha decay to the ground state as well as some low-lying excited…
A: The objective of this question is to show the ground state of California-252 (2 cm) undergoes alpha…
Q: Q3. Determine the oxidation number of the Chromium (Cr) in an unknown salt if electrolysis of a…
A: The objective of this question is to determine the oxidation number of Chromium (Cr) in an unknown…
Q: e. Please assign E or Z to each molecule CI Br Br OH CHO OCH3
A:
Q: 2. Cyclic voltammogram at a glassy carbon electrode for a 3 µM solution of Fe(II)heme in 0.10 M Bu 4…
A: The objective of this question is to show the cyclic voltammogram presents the current response over…
Q: The practical limit for ¹4℃ dating occurs when its activity falls to 0.20% of its original value due…
A: The aim of this question is to explain the carbon-14 period is based on the decay of 14C, which has…
Q: In a paragraph form, provide the experimental procedures with the laboratory equipments that needs…
A: The objective of this question is to explain the essential laboratory equipment are essential in…
Q: C)The line representing the gas phase in F.15 CTQ 2, should not be straight, especially over a…
A: The main aim of this question is to depicting the Gibbs free energy (G) and reaction rate (P) of H2O…
Q: Which of the following Newman projections represents (2S,3S)-dibromobutane? Br Br Br Br HC Br CH3 H…
A:
Q: H25.10 - Level 2 HomeworkUnanswered Due Today, 11:59 PM Sort the following carboxylic acid…
A: The reactivity of carboxylic acid derivatives towards nucleophilic substitution is determined by the…
Q: Explain the patterns and intensities of the isopropyl group in isopropyl iodide. CH3 CH3 CH-I What…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Why is it necessary to carry out the reduction of iron and then the titration, before goin on to the…
A: The objective of this question is to determination of iron in a sample through a redox titration.The…
Q: What is the concentration of tin ion in the anode if the Ecell for this reaction is 0.883 V and the…
A: The objective of the question is to find the concentration of tin ion in the anode given the Ecell…
Q: i. Why Redox active gels are cor ideal candidate to tune shapes, solubility, or display sol-togel…
A: The main aim of this question is to show the redox active gels are considered as ideal candidate to…
Q: НО. OH 1. 1-BuOK, I-BuOH 2. OSO4 3. NaHSO3, H2O НО. OH OH OH IV OH OH 애 Br OH
A: A question based on reaction of alkene. From the given reactant an alkene is produced, whose…
Q: You are analyzing the effluent from a lotion plant for parabens using HPLC. You have a working…
A: The objective of this question is to determination of the concentration of parabens in the…
Q: 100.0 mL of 0.50M OH basic solution is added to 2.00 L of a 0.250M acetate buffer at a pH of 5.0.…
A: Given,Molarity of OH- = 0.50 MVolume of OH- = 100.0 mLMolarity of acetate buffer = 0.250 Mvolume of…
Q: Draw the structure of the 6 nucleotide strand of alanine. Show all the atoms, bonds, and lone pairs.…
A: The objective of this yos to show the structure of the 6 nucleotide strand of alanine. It is an…
Q: Indicate the products that will be obtained from the following reactions and propose a mechanism. +…
A: The addition of halogens to alkenes involves an electrophilic addition reaction. Here's a detailed…
Q: Identify the reactants and products for the following reaction. Include predicted stereochemistry.…
A: Here we have to analyze the reaction between (R)-2-butylchloride and a nucleophile methyl sulfide.
Q: 1. The reversible reaction A B has the following rate equation: k2 dA dt (1) where A = A(t) and B =…
A: The aim of this question is to explain the reversible reaction A⇌B rate equation represented by a…
Q: Which of the following reactions is classified as a Williamson ether synthesis? O Nat 1. MCPBA 2.…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Show the detailed experimental procedures for the chemical synthesis of proteins with the…
A: The objective of this question is to show the α-Ketoacid-hydroxylamine binding is an effective…
Q: Identify the lonic and Covalent model for this compound, as well as the IUPAC name, and oxidation…
A: The objective of this question is to explain the given complex which show the features bridge…
Q: The rate of settling for 2.5 microgram powder in glycerin is 4.25*10^-8 How did we get this result?…
A: The settling rate of particles in a fluid is often described by Stokes' law, which states:Where:-…
Q: (A) 86α ULLE 58 72 60 Relative intensity 100- 15 m/z 20 40 80 100 114a Cattato co C3HCH₂N(CH3)…
A: Interpreting a mass spectrum involves identifying the main peaks and assigning them to specific…
Q: pentanol: 34.13 2,3-dimethyl-2-butanol: 44.06 If the concentration of pentanol in the unknown…
A: The objective of the question is to calculate the-The Response Factor.The area for .The area for…
Q: Once you have completed the second measurement, increase the volume to the max, 15.0 cm (15.0 nm on…
A: The aim of this question is to explain the pressure , volume and temperature of a gas, where moles…
Q: Choose a different family of natural products derived from the acetate pathway and propose how such…
A: The objective of this question is to show the different family of natural products derived from the…
Q: Draw an arrow-pushing mechanism for the Diels Alder reaction of anthracene-9- methanol and…
A: The main aim of this question is to show the reaction begins with a single cycloaddition forming a…
Q: A team mixed 14.00 mL of a 0.158 M solution of compound "A" with 18.00 mL of a 1.623 M solution of…
A: The objective of the question is to calculate the theoretical yield.
Q: What is the product? OA B. apl o O CI AICI3
A: Given,The reaction:
Q: %₂ ++ # # A) a (2s orbitals) B) a (2pz orbitals) C) TT (2px and 2py) D) T* (2px and 2py) E) o* (2pz)
A: We are given a molecular orbital diagram of O2We have to answer the LUMO in Molecular orbital…
Q: As the valence charge cloud gets larger, would it be easier/harder for an extra electron to move to…
A: The objective of this question is to show the two atoms approach each other, their potential energy…
Q: In the following reactions all reactants and products are gases. Which of the reactions, if carried…
A: In the following reactions all reactants and products are gases
Q: P7.B* Sketch a MO energy level diagram for H2– in the ground state, show electrons by spin-arrows.…
A: The objective of this question is to show the energy level diagram of its molecular orbital in the…
Please don't provide handwriting solution
Step by step
Solved in 3 steps with 1 images

- How many grams of Chromium (III) oxide are produced with 2.0g of ammonium dichromate? (balanced equation: (NH4)2Cr2O7 -> Cr2O3 + 4H2O + N2)A 0.4044 g solid organic sample containing covalently bound bromide and no otherhalogens was placed in a porcelain crucible with about one gram of fresh sodium metal. Ina process known as sodium fusion, the mixture was heated in a furnace to 450°C whichcharred and vaporized the organic portion of the molecule and converted the covalentbromide into sodium bromide. Excess sodium was decomposed by adding small portionsof water which also dissolved the sodiumbromide. The clear solution was quantitatively transferred to a beaker, acidified with dilutenitric acid, and diluted to about 50 mL. A 6 mL quantity of 0.1 M AgNO₃ was added to thesolution andthe mixture heated to about 60°C for an hour to age and digest the precipitate. After filtering,the mass of the silver bromide produced was determined to be 37.8 mg. What is thepercentage by mass bromine in the organic compound? (Ag=107.868, N=14.0067,O=15.9994, Br=79.904)formula for a compound is given as C₂H₁₂O₆. This is a (an)
- If cobalt(II) sulfate is heated too strongly, the following reaction will occur CoSO4(s) à CoO(s) + SO3(g) If you are heating a sample of CoSO4·6H2O and this reaction occurs along withdehydration, what will happen to the experimental percent water? Explain your answer.The nurse needs to prepare a medicine that would help the x-ray images of the gastrointestinal. What chemical test would the nurse perform to determine whether the barium compound to use is BaSO4(s) or BaCO3(s)?Consider the series of reactions to synthesize the alum (KAl(SO4 )2 · xH2O(s)) from the introduction. (a) Assuming an excess of the other reagents, from one mole of aluminum Al (s), how many moles of alum will be produced? (b) Assuming an excess of the other reagents, from one mole of potassium hydroxide KOH, how many moles of alum will be produced? (c) Assuming an excess of the other reagents, from one mole of sulfuric acid H2SO4 , how many moles of alum will be produced? (d) If you start the synthesis with 1.00 g of Al, 40.0 mL of 1.50 M KOH, and 20.0 mL of 9.00 M H2SO4 , which of the three will be the limiting reagent? (e) Assuming that the product is anhydrous (that there are no waters of hydration), calculate the theoretical yield of alum, in grams, based on the amounts of reagents in part (d). 3. Consider the nickel salt: (NH4 )2Ni(SO4 )2 ·y H2O (Ammonium Nickel Sulfate Hydrate), where y is the number of coordinated waters. (a) Assuming that the product is anhydrous (y = 0),…



