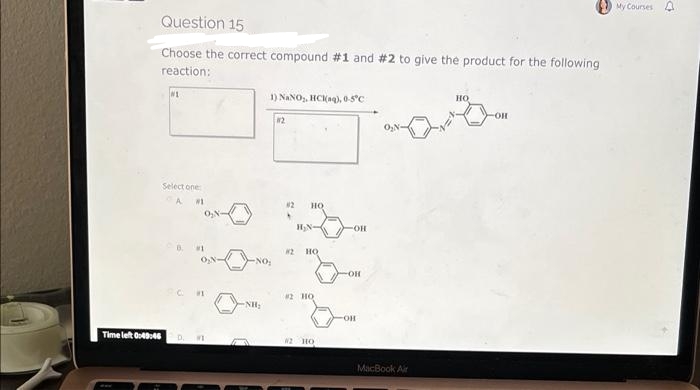
Question 15 Choose the correct compound #1 and #2 to give the product for the following reaction: NE 1) NaNO₂, HCl(aq), 0-5°C -OR 82 Select one A#1 #1 #1 Time left 0:49:46 D #1 NO₂ NH₂ 82 HQ H₂N- HỌ #2 HO #2 HO -OH -OH -OH
Q: sider the following reaction at 298K. (aq) + Cr²+(aq)→→→→→→ Ag(s) + Cr³+ (aq) standard Gibbs free…
A:
Q: Consider the following reaction: 2- bromooctane reacted with Sodium tert-butoxide complete reaction…
A: 2-bromooctane + sodium tert-butoxide --> ->Sodium tert-butoxide has more basic character than…
Q: 16. What will be the final temperature of a 584−g sample of water, initially at 15.0 °C, after 53.4…
A: 584 g sample of water, initially at 15.0 °C. 53.4 KJ of heat have been added to it.Here we have to…
Q: Provide all products AND mechanisms for the following reactions (no transition states or energy…
A: Given reactions are : Provide all products and mechanism of the given reactions = ?
Q: Suppose you have a soil that is made up of 10% organic matter with CEC = 200 cmol/kg, 40% kaolinite…
A: Given: A soil with- 10% organic matter with CEC = 200 cmol/kg 40% kaolinite with CEC = 10 cmol/kg…
Q: onsider the following reaction at 298K. Fe³+(aq) + Cd(s)→→→→→2Fe²+ (aq) + Cd²+ (aq) he standard…
A:
Q: A brick has a mass of 4.0 kg and the Earth has a mass of 6.0 × 10²7 g. Use this information to…
A:
Q: Which of the following thermochemical equations represents the formation of liquid sulfuric acid?…
A:
Q: Solid ammonium chromate is slowly added to 75.0 mL of a calcium acetate solution until the…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Provide the reagents necessary to carry out the following synthesis Drag the appropriate labels to…
A: Complete reaction step is given below in next step
Q: Click on the part of tetrahydrofolate that is functionalized to create an activated carrier of…
A: The function of tetrahydrofolate is to create an activated carrier of various carbon groups.
Q: How many electrons would the most stable cation of Ca have
A:
Q: What are the major differences between GC and HPLC, discus the types of samples among other…
A: GC is typically used to measure oils, organic compounds, air samples, toxins and drugs (both…
Q: Ca²+ (aq) + 2e → Ca(s) OA.0.21 V OB. -0.47 V OC -0.21 V OD.0.47 V OE. 0.42 V A Moving to another…
A:
Q: Enter electrons as e. Use smallest possible integer coefficients. If a box is not needed, leave it…
A:
Q: What is observed with the Ksp of of AgCl and Ag2Co3 at two different temperatures?
A: Ksp is the solubility product as it is the product of the solubilities of the ions in moles per…
Q: Balance the reaction between CrO42- and Hg to form Cr³+ and Hg2+ in acidic solution. When you have…
A: Given in following question a reaction unbalanced it's balanced and howbmany electron transfer in…
Q: Find the AH for the reaction below, given the following reactions and subsequent AH values: C3H8(g)…
A:
Q: What is the pH and pOH concentration of a 0.072 M of methylamine? Write the ionization equation and…
A: Given, Concentration of the methylamine = 0.072 M Kb for methylamine = 4.6x10-4 pH of the solution…
Q: Which of the following is NOT expected to result in an efficient crossed aldol condensation…
A:
Q: Y Salt bridge A+ B- Y2+ - the half-cell containing Y is the , the half-cell containing X undergoes…
A: In the galvanic cell, anode is negative charged electrode and oxidation takes place. And cathode…
Q: H-CI *** →
A: 1,4- product is formed in thermodynamic controlled product formation case. Where more stable alkene…
Q: Consider the following reaction: 2Sb2S3 (s) + 6PbO2 (s) → 2Sb2O3 (s) + 6PbS (s) + 30: (g) a)…
A:
Q: 29. A 275 mL sample of gas is kept at 25°C. What volume will the gas occupy at 100.°C?
A: For a definite amount of gas, at constant pressure, the volume of the gas increases with an increase…
Q: 2 OF2(g) + 2 S(s) → SO2(g) + SF4(g) OF2(g) + H2O(l) → O2(g) + 2 HF(g) ΔH = – 78.5 kJ…
A:
Q: The free energy change for the following reaction at 25 °C, when [Hg2+] = 1.19 M and [Zn²+] =…
A: Answer: Relation between Gibbs free energy change and cell potential is shown below: ∆G=-nFEcell…
Q: 5) C,H,X in this compound MASS SPECTRUM 100 80 60 40 20 0.0+ 20 40 60 80 100 m/z NIST Chemistry…
A: Note : M: M+2 ration is 3:1. Therefore the compound is having chlorine atom. m/z = 77 belongs to…
Q: To prepare the buffer, a total volume of 300 mL is required. The stock solution of the weak base is…
A: In the given question we have to calculate the required concentration of weak base to prepare a…
Q: How many liters of 0.88 m solution can be made with 25.5 grams of lithium fluoride
A:
Q: How many valence electrons do you have to account for when drawing the Lewis dot structure of PCl5?
A: Valence electron of phosphorus is five and valence electrons of chlorine is seven
Q: immersed in a solution where [Cu²+] = 1.00 M, is 0.340 V. The positive voltage indicates that the…
A: The standard reduction potential for the copper is given to be 0.340 V. We are required to find out…
Q: Using the table of the weak base below, you have chosen Methylamine as your weak base in the buffer…
A: Given: pH of buffer = 9.75
Q: Molten CaCl₂ can be electrolyzed to form metallic Ca. For what duration should an electrolysis be…
A:
Q: Half-reaction E° (V) Br₂(1) + 2e¯ →→→→→→ 2Br¯(aq) 1.080V Cu²+(aq) + 2e¯ →→→→ Cu(s) 0.337V Mn²+(aq) +…
A: Oxidizing agent is the species that oxidizes the other species and itself gets reduced. Reducing…
Q: Balance the reaction between BrO3 and I to form Br₂ and I₂ in basic solution. When you have balanced…
A: Here we are required to balance the redox reaction in basic condition
Q: What is the Common Crystal Habits of the following: - Pyrope - Almandine - Spessartine - Uvarovite -…
A:
Q: An acid spill in a pond needs to be cleaned up with a base. a) Which chemical should be considered…
A: Given: An acid spill in a pond needs to be cleaned up with a base.
Q: Molten CaCl₂ can be electrolyzed to form metallic Ca. For what duration should an electrolysis be…
A: Faraday's first law :- It states that, the amount of the substance deposited at the electrode is…
Q: calculate the pH of a solution that is 1.00 M CH3COOH (ka = 1.0 x 10^-5) and 1.00 M CH3COONa
A: Given: Concentration of weak acid CH3COOH = 1.00 M And the concentration of CH3COONa = 1.00 M
Q: 24. A deoxyhemoglobin molecule (molar mass 65,000 g) can bind up to four oxygen molecules. In a…
A:
Q: Consider the galvanic cell based on the following half-reactions: Mn²+ +2e →Mn = -1.18 V Fe²+ + 2e →…
A: Here we are required to find the standard potential of the cell reaction along with the equilibrium…
Q: The mass of a beaker is 5.333g. After 5.00mL of a concentrated hydrochloric acid solution is…
A:
Q: 28. if 5 moles of NaCl is added to a 1 liter of pure water what will be the percent by mass of the…
A:
Q: 2 NO (g) + O₂ (g) --> 2 NO2 (g) The proposed mechanism for the reaction above is 1) NO (g) + NO (g)…
A: Answer: For a multistep reaction, slowest step is always the rate determining and since, over here…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: The compounds given are BaCO3, CaI2 and CuBr.
Q: Calculate the pH of 0.10 M solution of hypochlorous acid, HOCl, Ka = 2.9 x 10-8 (Weak acid) HOCl ⇌…
A: In multiple questions we solve only first question according to the Bartleby guideline. First find…
Q: What is the emf of a galvanic cell consisting of a Cd²+/Cd half-cell and a Pt/H/H₂ half-cell at the…
A:
Q: 5. Propose a structure for a compound, with molecular formula C8H19N, that fits the following…
A: Molecular formula C8H19 N Formula for degree of unsaturation of a molecule CnHx Ny = n+1-x2-y2 Now…
Q: Choose the correct starting material and reagents for this transformation using a Diekmann…
A: Note : Intramolecular Claisen condensation is called Dieckamann cyclization. (see below for…
Q: 14. Calculate and compare [H₂O"] and pH for a 0.10 M solution of HCI (a strong acid) and a 0.10 M…
A: Acid are compound which releases hydronium ion when dissolve in water We are required to compare…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Hello, I was hoping for some help with the prelaboratory questions for Analytical Chemistry. I am pretty sure I got number one, but I was very confused about the "second endpoint" calculations and I am pretty sure I have calculated the percentage wrong. These are the equations we are given to work with and the questions are below. Thanks in advance! Eq. 2 CO32- + H+ Û HCO3- (bicarbonate ion) Eq. 3 HCO3- + H+ Û H2CO3 (carbonic acid) Eq. 4 However: H2CO3 (aq) Û CO2 (g) + H2O How much concentrated HCl acid is required to make 0.500 L of a 0.100 M HCl solution? One can calculate the Molarity (M) of the HCl from the weight percentage on the reagent bottle (which is usually 37.0% (w/w) with a density of 1.19 g/mL). A weight of 1.300 g of sodium carbonate (MW = 105.989 g/mol) is dissolved in 250.0 mL water. If a 25.00 mL aliquot of the sodium carbonate solution is placed into an E-flask and titrated with 0.1034 M HCl, what should be the volume of HCl to reach the second…Hydrazine N2H4can interact with water in two stepsN2H4(aq) + H2O (l) ↔N2H5+(aq) + OH-(aq) Kb1= 8.5 x 10-7N2H5+(aq) + H2O (l)↔N2H62+(aq) + OH-(aq) Kb2= 8.9x 10-16a)What is the concentration of OH-; N2H5+; and N2H62+in a 0.010 M solution of hydrazine?b)What is the pH of the 0.010 M solution of hydrazine?(please see pictures attached for reference. I tried my best) 27. Dissolved dioxygen in water samples can be determined by the following equations:2MnSO4(aq) + 4NaOH(aq) + O2(g) --> 2MnO2(s) + 2Na2SO4(aq) + 2H2O(l)2MnO2(s) + 2H2SO4(aq) + 2NaI(aq) -->MnSO4(aq) + I2(aq) + Na2SO4(aq) + 2H2O(l)I2(aq) + 2Na2S2O3(aq) --> Na2S4O6(aq) + 2NaI(aq)A water sample from the local river was treated according to the first two equations above. Exactly 200.00 mL ofthis treated water required 8.32 mL of 0.0250 M Na2S2O3 to titrate to the endpoint. Calculate the concentration ofdissolved oxygen in the water sample.A) 1.04 x 10-3MB) 2.08 x 10-4MC) 5.20 x 10-4MD) 8.67 x 10-7ME) 4.33 x 10-7M 28) How many neutrons are needed to initiate the fission reaction shown? 23592 U + ? 10 n --> 13956 Ba +9436 Kr+ 310n a) 0 b) 1 c) 2 d) 3 e) 5 20. Consider the substances H2O, KI, H2S, CH4. Which answer has the substances arranged in order ofDECREASING boiling point?a) KI > H2O > CH4 > H2S b) KI…
- What reagent/s is/are needed to confirm the identity of the cation in the diagram? a. acetic acid b. Ba(NO3)2 c. 6M H2SO4 d. excess NaOH e. excess ammonia f. ammonium oxalate g. nitric acid h. Fe(NO3)3 i. K4Fe(CN)6 j. K3Fe(CN)6 k. ammonia l. MgCl2 m. potassium permanganate n. 18M H2SO4 o. KSCN p. NaOH q. ferrous sulfateWhy is H2SO4 a weaker acid than HClO4? I. S is less electronegative than Cl. II. H2SO4 has fewer terminal O atoms than HClO4. III. The S–H bond is less polar than the Cl–H bond. Select one: a.I only b.I and II only c.I, II, and III d.II onlyWhat fraction of iminodiacetic acid is in each of its four forms ( H3A H2A− , HA2− , A3− ) at pH 3.651 ? The p?a values for iminodiacetic acid are 1.85 ( p?a1 ), 2.84 ( p?a2 ) and 9.79 ( p?a3 ). ?H3A= ? α H2A = ? ?HA2−= ? αA3− = ?
- Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. Cs2CO3(s) = 2Cs+(aq) + 3CO(aq) Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. CH3OH(aq) = Complete the balanced dissociation equation for the compound below in aqueous solution. If the compound does not dissociate, write NR after the reaction arrow. Mg(OH)2(s) =Select every equation that is expected to favor the products. Question options: SCN- + HSeCN HSCN + SeCN- PH2- + S-2 HS- + PH-2 HCF3 + CCl3- CF3- + HCCl3 H2XeO4 + XeO4-2 2 HXeO4-1 NO2- + HNO NO- + HNO2Ques 3&4 do any 1 thank you
- Given the table below, what is the most probable identity of the acid?Acid KaHypochlorous acid 3.0 x 10⁻⁸Nitrous acid 7.1 x 10⁻⁴Hydrogen cyanide 6.2 x 10⁻10Propionic acid 1.3 x 10⁻⁵Consider the following reaction: X-SH ⇌ X-S- + H+ The pKa for the sulfhydryl (thiol) group (SH) is 8.37. Choose any/all statements that apply: X-SH is a base. X-S- is the conjugate base of X-SH. H+ is a proton acceptor. X-S- is the conjugate acid of X-SH. At neutral pH, the equilibrium lies such that X-S- is the predominant species.Hello. Please help me with a, b, and c. Handwritten solutions are prefered for more clear understanding Liquid ammonia (anhydrous NH3(l)) is often used as a solvent. Like water, ammonia undergoes autoionization by the transfer of a proton: 2NH3 = NH4+ + NH2- K = 1×10-33 at -50°C Since ammonia is better proton acceptor than water, the ionization of acids is relatively enhanced in liquid ammonia. For example, acetic acid is a strong acid in liquid ammonia. 1a) Estimate [NH4+] (mol/L) in pure (i.e. neutral) liquid ammonia at -50°C. 1b) Calculate [NH4+] (mol/L) in a 5.33×10-1 M solution of acetic acid in liquid ammonia at -50°C. 1c)Estimate [NH2-] (mol/L) in a 5.33×10-1 M solution of acetic acid in liquid ammonia at -50°C.

