Question 15 The equivalence point of a titration of 350.0 mL of 0.00016 M weak acid with 0.00082 M strong base is reached when 480.0 mL of strong base is added. What is the pKa of the weak acid? You Answered 1.03 Correct Answers 4.32 (with margin: 0.02)
Question 15 The equivalence point of a titration of 350.0 mL of 0.00016 M weak acid with 0.00082 M strong base is reached when 480.0 mL of strong base is added. What is the pKa of the weak acid? You Answered 1.03 Correct Answers 4.32 (with margin: 0.02)
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.28QAP
Related questions
Question
help me find out Pka
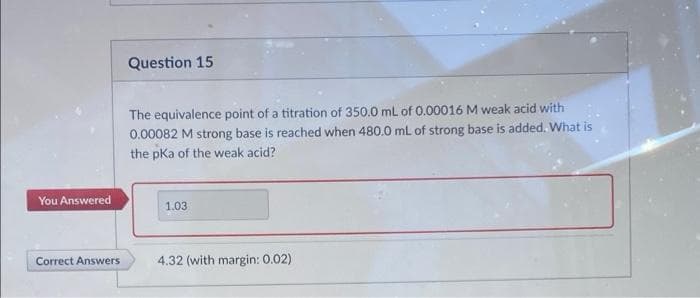
Transcribed Image Text:Question 15
The equivalence point of a titration of 350.0 mL of 0.00016 M weak acid with
0.00082 M strong base is reached when 480.0 mL of strong base is added. What is
the pKa of the weak acid?
You Answered
1.03
Correct Answers
4.32 (with margin: 0.02)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



