Question 15 Which among the following is an equivalent of 0.01%? O 100 ppm O 100 ppb O 0.000001 ppm O 0.000001 ppb O 0.0001 ppm D Question 16 Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits. O 13.58 g/cm' O 13.66 g/cm O 183 g/cm O 0.0732 g/cm O 0.073 g/cm Question 17 often shinned in 55-gallon drums. What is the weight in pounds of a 55-gallon drum if the density of the waste is 1.5942 g/cm7 (454 g=1 pound; 0.9463 L- I quart; 4 quarts - I gallon)
Question 15 Which among the following is an equivalent of 0.01%? O 100 ppm O 100 ppb O 0.000001 ppm O 0.000001 ppb O 0.0001 ppm D Question 16 Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits. O 13.58 g/cm' O 13.66 g/cm O 183 g/cm O 0.0732 g/cm O 0.073 g/cm Question 17 often shinned in 55-gallon drums. What is the weight in pounds of a 55-gallon drum if the density of the waste is 1.5942 g/cm7 (454 g=1 pound; 0.9463 L- I quart; 4 quarts - I gallon)
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter1: The Nature Of Chemistry
Section: Chapter Questions
Problem 18QRT
Related questions
Question
I'm taking a practice test and I'm having some trouble and was hoping you could help. This is not graded I just need help
See attached:
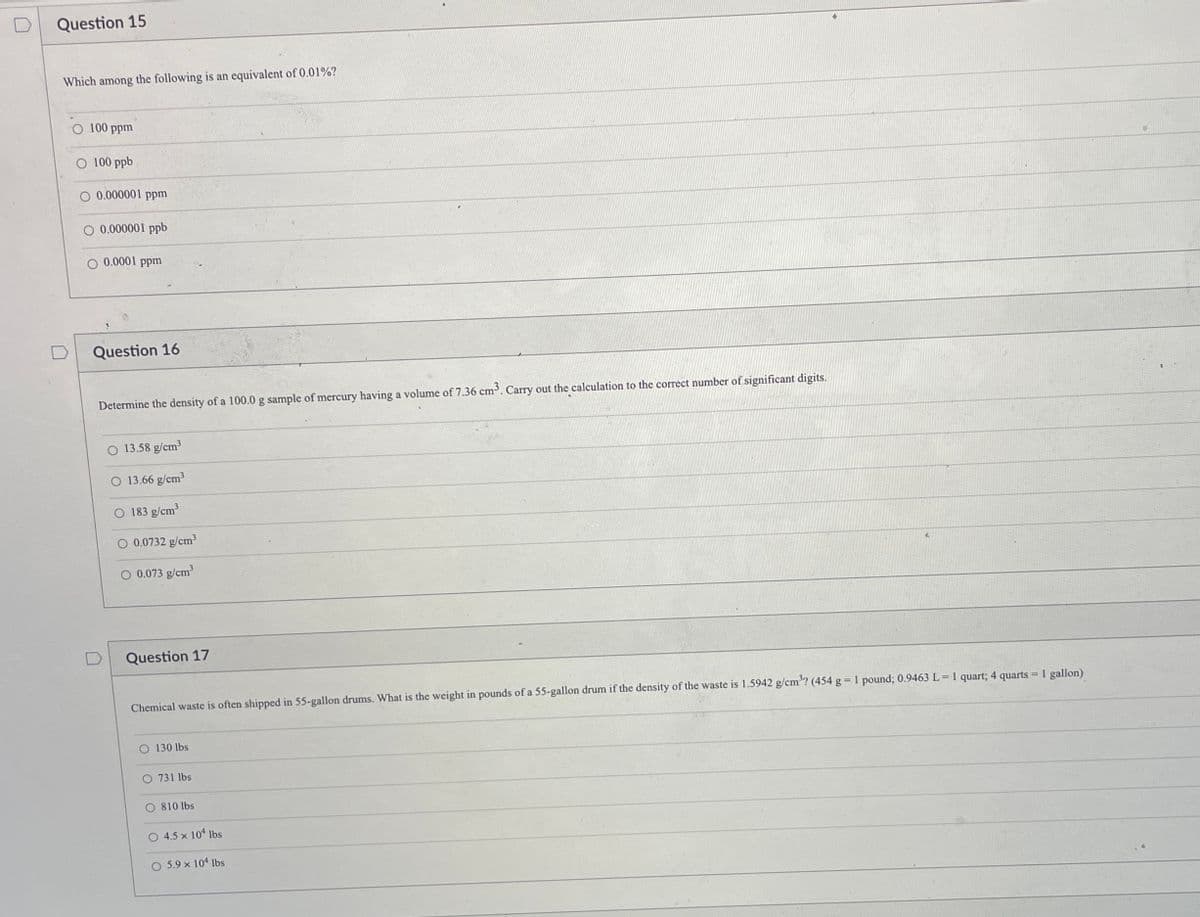
Transcribed Image Text:Question 15
Which among the following is an equivalent of 0.01%?
100 ppm
O 100 ppb
O 0.000001 ppm
O 0.000001 ppb
O 0.0001 ppm
Question 16
Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits.
13.58 g/cm?
13.66 g/cm3
183 g/cm
O 0.0732 g/cm3
O 0.073 g/cm
Question 17
Chemical waste is often shipped in 55-gallon drums. What is the weight in pounds of a 55-gallon drum if the density of the waste is 1.5942 g/cm'? (454 g 1 pound; 0.9463 L = 1 quart; 4 quarts = 1 gallon)
O 130 lbs
O 731 lbs
O 810 lbs
O 4.5 x 104 lbs
O 5.9 x 104 lbs
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning