Question 18 In GC experiment, a standard solution containing 0.147 grams ethanol gave a peak area of 9,930. A 0.983 grams of an unknown sample containing ethanol gave a peak area of X 6.382. Calculate the mass percent of ethanol in the unknown sample. Selected Answer:o 14.7% Answers 14.7% 0.83 % Zero %
Question 18 In GC experiment, a standard solution containing 0.147 grams ethanol gave a peak area of 9,930. A 0.983 grams of an unknown sample containing ethanol gave a peak area of X 6.382. Calculate the mass percent of ethanol in the unknown sample. Selected Answer:o 14.7% Answers 14.7% 0.83 % Zero %
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 7P
Related questions
Question
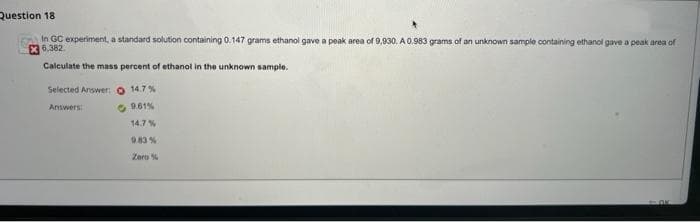
Transcribed Image Text:Question 18
In GC experiment, a standard solution containing 0.147 grams ethanol gave a peak area of 9,930. A 0.983 grams of an unknown sample containing ethanol gave a peak area of
X 6.382.
Calculate the mass percent of ethanol in the unknown sample.
Selected Answer:O 14.7%
Answers
14.7%
0.83 %
Zero %
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

