Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.12QAP
Related questions
Question
Transcribed Image Text:9 HW Chapter 19 - Selected Exercises
Question 2 of 8
< >
0.6/1
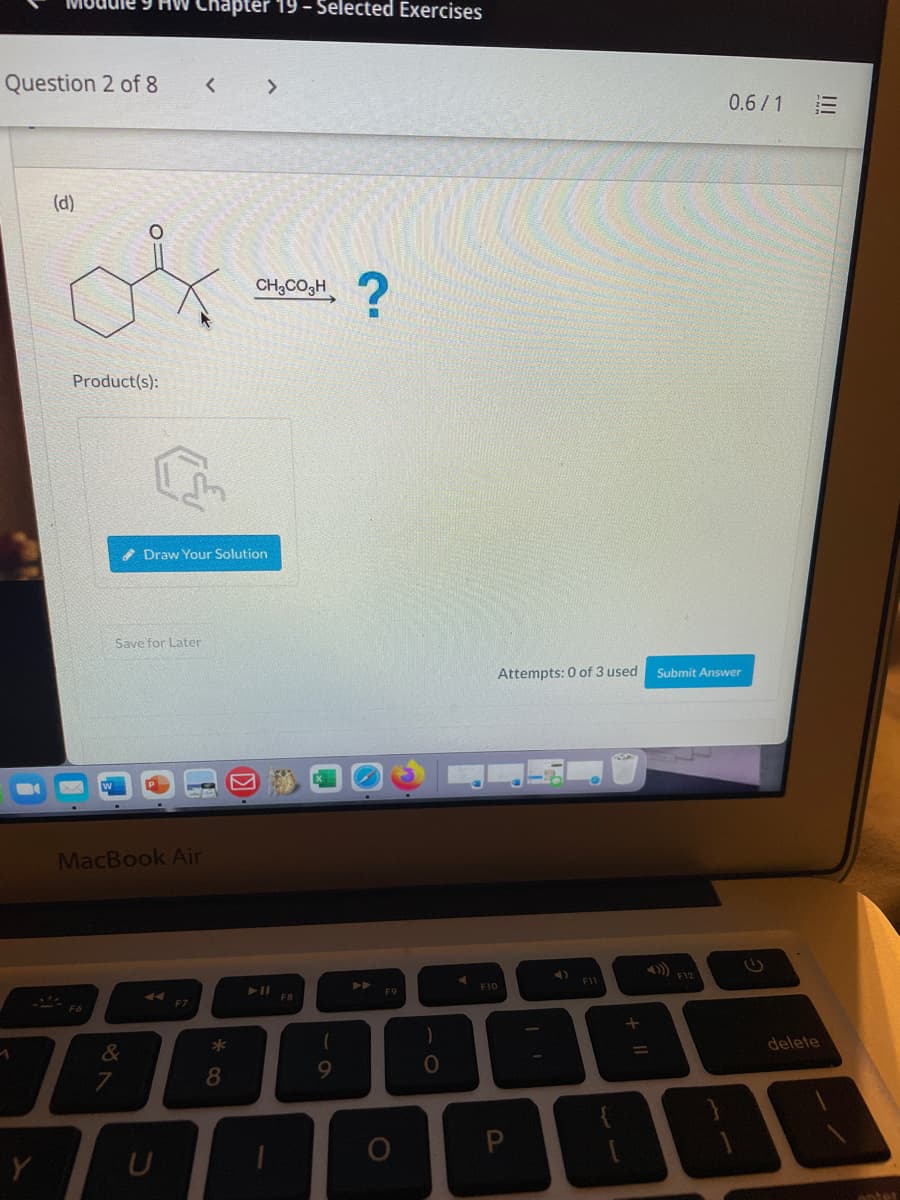
(d)
CH,CO3H
Product(s):
Draw Your Solution
Save for Later
Attempts: 0 of 3 used
Submit Answer
MacBook Air
A) F12
F11
F10
F9
F7
delete
&
8
Y
enter
II
Transcribed Image Text:e Module 12 HW C
NWP Assessme:
el Module 9 HW C
WWP Assessme
Module 11 HW
MD.edu
A https://education.wiley.com/was/ui/v2/assessment-player/index.html?launchld39010073f-214
HW Chapter 20 – Selected Exercises
Question 3 of 8
< >
View Policies
Current Attempt in Progress
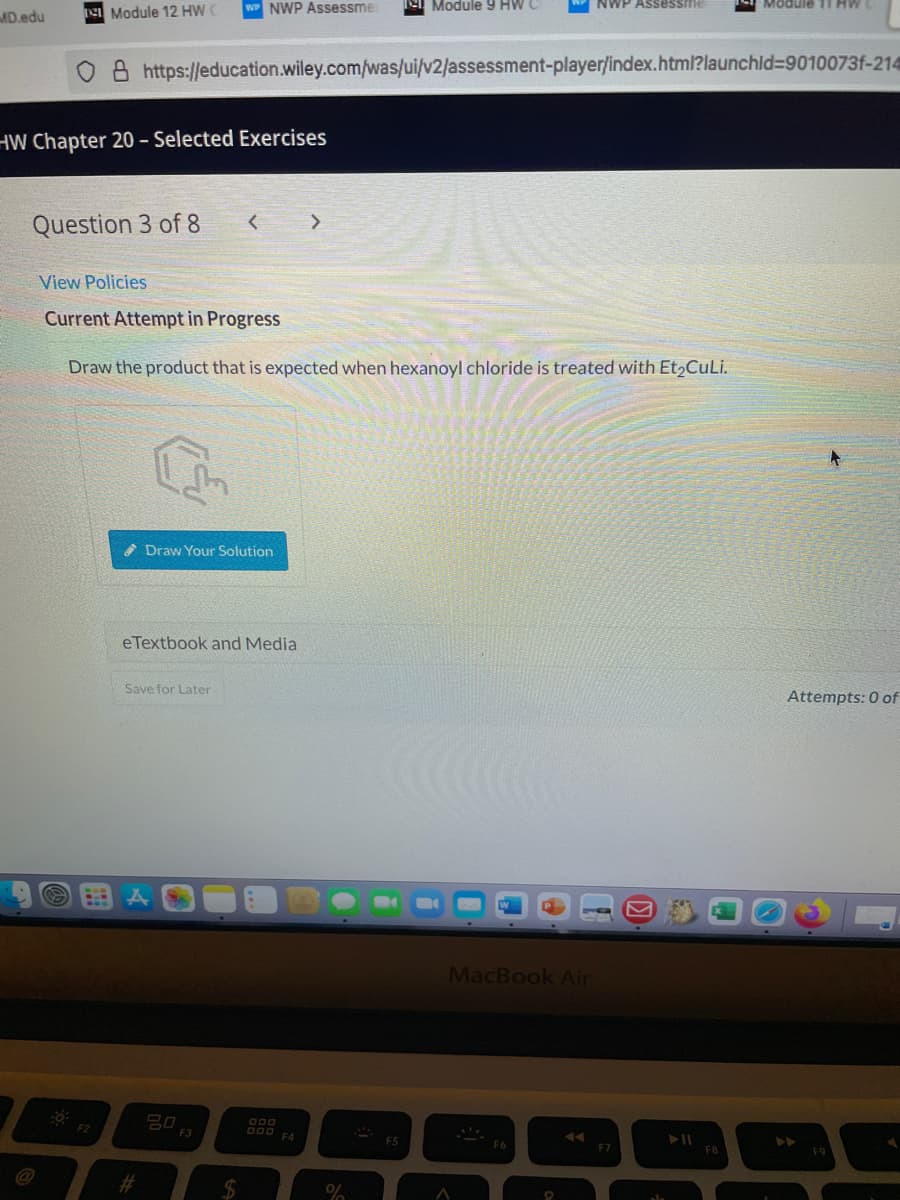
Draw the product that is expected when hexanoyl chloride is treated with Et2CuLi.
* Draw Your Solution
eTextbook and Media
Save for Later
Attempts: 0 of
MacBook Air
O00
F3
000 A
F7
2#3
%24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

