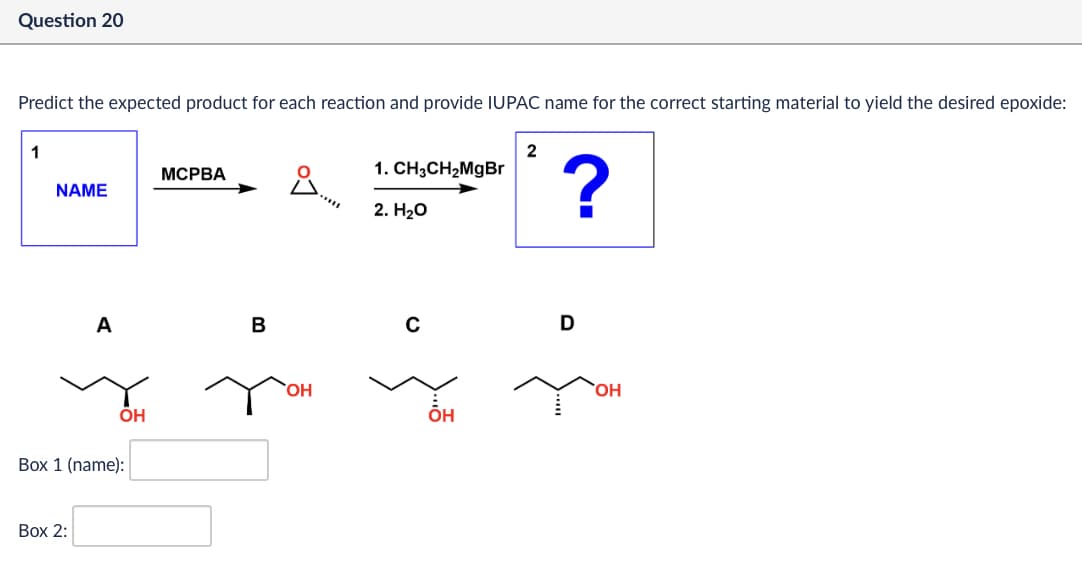
Question 20 Predict the expected product for each reaction and provide IUPAC name for the correct starting material to yield the desired epoxide: 1 2 MCPBA NAME ů 1. CH3CH2MgBr ? 2. H₂O A B OH Box 1 (name): Box 2: OH C D OH OH
Q: 9. Draw the following chemical reactions. a) cyclopentylamine + water b) cyclopentylamine + butanoic…
A: Step 1: Step 2: Step 3: Step 4:
Q: Complete the following reaction: CHO H- OH H H OH + Cu2+ OH CH₂OH
A: This is called Benedict's test which is used to detect reducing sugars.Reducing sugars are…
Q: Please explain this synthesis step by step.
A: Step 1: Step 2: Step 3: Step 4:
Q: Calculate internal energy change. A gas is compressed and during this process the…
A:
Q: Complete the following nuclear equations. Write the mass number, atomic number, and symbol for the…
A: (a) 178O + 11p → 147N + 42He(b) 2713Al + 10n → 2411Na + 42He(c) 74Be + 10n → 73Li + 11H(d) 136C…
Q: Rimantadine is an antiviral drug used to treat people infected with life-threatening influenza…
A:
Q: The corrected cell potential for the following galvanic cell was determined to be (1.62x10^0) V.…
A:
Q: In the following FTIR Spectrum, what functional group can you recognize?
A: Step 1: Step 2: Step 3:
Q: In aqueous solution the Ag ion forms a complex with two cyanide anions. Write the formation constant…
A: Step 1:Step 2:Step 3:Step 4:
Q: The concentration of copper in a sample of sea water is determined by anodic stripping voltammetry…
A: Peak currents in anodic stripping voltammetry are a linear function of concentration We also know…
Q: ABCD Question 6 Please predict the product for each of the following reactions. Make sure to clearly…
A: Two major steps occur in the ring opening of epoxide in acidic conditions.First, epoxide gets…
Q: 53) The valence electron configurations of several atoms are shown here. How many bonds can each…
A: Step 1:Valance electron:The electrons which are present in the outer most energy level are known as…
Q: A tank contains 330 gallons of oil. This is the equivalent of 4.83×1010 J of energy. How many kcal…
A: Approach to solving the question:Please see attached photos for detailed solutions. Thank you.…
Q: Predict the major product for the following reaction. Ans: OH H3O+
A: Step 1 :
Q: None
A: The IUPAC name for benzyl benzoate is derived as follows:Benzyl: This part of the name refers to the…
Q: None
A: Due to heating 1,3-dicarboxylic acid is converted into monocarboxylic acid due to decarboxylation of…
Q: Draw a structural formula for the alkene you would use to prepare the alcohol shown by…
A: Step 1: Step 2: Mechanism Step 3:Step 1: HydroborationThis step involves the reaction of the alkene…
Q: Name these organic compounds please:
A: Step 1: Notice that all the given compounds only have single bonds. This means that all three…
Q: The main hormonal organs of the gonadal system are: Question 9 options:…
A: The gonadal system refers to the reproductive system's organs that are responsible for producing…
Q: Please provide a mechanism for the reaction with P-bromophenacyl ester. The unknown is 3-pentanone.
A: Mechanism of the reaction with p-bromo phenacyl ester (PBPh), where the unknown is 3-pentanone:1.…
Q: The standard cell potential (E°cell) for the reaction below is +0.63 V. The value of ΔG° for the…
A: Step 1: The relationship between cell potential (E°cell)and Gibbs energy (ΔGo) can be expressed by…
Q: A hot 120.8 g lump of an unknown substance initially at 176.2 °C is placed in 35.0 mL of water…
A: For No. 1: The amount of energy in the form of heat that is released by the hot lump of unknown…
Q: Electronic State Χ(ΙΣ) V/cm-1 Vex/cm¹ B./cm-1 А (П1) 384.29 212.3 1.501 2.39 0.1141 0.0843 D./cm-1…
A: I can't access the specific image you sent, but based on your description of the circuit being an…
Q: What is the enegry numbers for the table?1 calorie (cal) = 4.184 J1 kWh = 3.600 × 106 J
A: Please see the attached image for the solution. If there are queries,please do not hesitate to ask.…
Q: Part a and b please
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following carbocations is/are likely to rearrange? IV I II Ob. III Oc II and V Od IV
A: Here, we are aware of the following carbocation stability: Whereas secondary carbocation is more…
Q: Drawing Lewis Structures For each molecule, calculate the total number of valence electrons. Then,…
A: Detailed explanation:Examples:H2SO4
Q: Please don't use hend raiting
A: if mechanism will require I will send it .
Q: None
A: Certainly, I can assist you with calculating the standard Gibbs free energy of reaction (ΔG°rxn) for…
Q: None
A: Step 1: Given that Mass = 79g Volume = 47 cm3 Density can be calculated as Density = Mass/Volume…
Q: Predict the products of the following reactions. Show stereochemistry when applicable.
A: The second reaction, the addition may occur via syn or anti pathway. So both cases are possible
Q: None
A: In urea (H₂NCONH₂), the carbon atom is situated within the central molecular structure, forming…
Q: None
A: Step 1:Assign priority to the two substituents attached to each carbon atom of the double bond. Use…
Q: Show work, thank you!
A: Please note that this is a theoretical calculation and actual results may vary based on experimental…
Q: Question 3
A: A molecule with sp³d hybridization typically has a trigonal bipyramidal electron pair geometry. This…
Q: ABCD Question 1 Please predict the product for each of the following reactions. Make sure to clearly…
A:
Q: Show work, thank you!
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: A galvanic cell is composed of these two half-cells, with the standard reduction potentials shown:…
A: Given:Zn2+ (aq) + 2e- <---> Zn (s) -0.76 volt Cd2+ (aq) + 2e-…
Q: What would the potential of a standard hydrogen electrode (S.H.E.) be under the following…
A: Here's how to find the potential of the SHE under these conditions:Standard Hydrogen Electrode…
Q: Indicate the products A and B that are obtained in the following reactions: OH CH3 + HOCH2-CH₂OH +…
A:
Q: Problem 79 of 80 Submit Curved arrows are used to illustrate the flow of electrons. Using the…
A:
Q: I would appreciate any help on how to solve this problem :)
A: Given: masNO2=10.2g;MN=14.01g/mol;MO=16.00g/mol2NO2(g)→N2O4(g)ΔH°=−55.3kJStep 1: Is the…
Q: Question 12 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: Plz explain, no handwritten answer
A: Step 1:To solve this problem, we can use Henry's Law, which states that the amount of gas dissolved…
Q: Please give a detailed stepwise mechanism for the following reactions. All arrows, charges, and…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Question 15 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: Step 1: Step 2: Step 3: Step 4:
Q: Major organic product predict
A:
Q: The following IR spectrum is for the molecule 3-pentanone. Please label and provide a table for the…
A:
Q: Draw the most stable chair conformation of each of the following compounds. (a)cis-1,1,4 -…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 8: A sample consisting of 1.00 mol Ar is expanded isothermally at 0°C from 22.4 dm³ to 44.8…
A:
Step by step
Solved in 2 steps with 1 images

- (please answer all questions) 1) Predict the products from reaction of 1-hexyne with thesereagents: a) One equivalent HBr b) One equivalent Cl2 c) H2 , Lindar Catalyst d) NaNH2 in NH3 , then CH3BrI recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.Could you please help me with this question I am very stuck. Give the major organic product of each reaction of methyl pentanoate with the given 6 reagents under the conditions shown. Do not draw any byproducts formed. (see photo) a. Reaction with NaOH,H2O heat; then H+,H2O b. Reaction with (CH3)2CHCH2CH2OH(excess), H+ c. Reaction with (CH3CH2)2NH and heat. d. Reaction with CH3MgI(excess), ether; then H+/H2O e. Reaction with LiAlH4, ether; then H+/H2O f. Reaction with DIBAL (diisobutylaluminum hydride), toluene, low temperature; then H+/H2O
- In the synthesis of methyl m-nitrobenzoate from methyl benzoate using HNO3 and H2SO4 via EAS the product is washed with ethanol to remove impurities. By impurities do they mean o-nitrobenzoate and methyl p-nitrobenzoate? How come ethanol can remove o-nitrobenzoate and methyl p-nitrobenzoate, but has no effect on m-nitrobenzoate? Thank you.redraw the following reaction mechanism with benzene instead of isoborneol. it's an oxidation mechanism from OH to ketone. please include the arrow pushing.QUESTION 4Predict the products(s) from the reaction of 1-hexyne with each of the following reagents:a) 1 mole of HBrb) H2O, H2SO4, HgSO4c) 2 moles of Br2d) Excess Cl2e) 2 moles of HCl with H2O2f) 1 mole of HCl
- (a) What happens when CH3—O—CH<sub3 is heated with HI?(b) Explain mechanism for hydration of acid catalyzed ethene :CH2 = CH2 + HzO CH3—CH,—OHEXP VIDEO https://www.youtube.com/watch?v=9Wxxv7kwXlA&list=PLE-217H0ao60Uzpzj-FNP_0ScVp5SZB7j&index=8 1.Pre lab question What are the functional groups in your substrate? -Nitrate -Nitrate and acetate -Amide and ether -Benzene ring b.The stable reaction intermediate in this reaction is Nitronium ion Benzonium ion Carbocation Carbanion c.How would you distinguish between 2-nitrophenacetin and 3-nitrophenacetin? -Color difference -Melting point difference -Difference in Molecular weight -They cannot be differentiated. dAn ether group is -Ortho para activating -Meta activating -Ortho para deactivating -ring activator eThe catalyst in this experiment was -Glacial acetic acid -Nitric acid -Sulfuric acid -Ethanol fThe chemical waste from this experiment should be discarded in the -Sink -Halogenated waste container -non-halogenated waste container -Heavy metal waste container. gWhat is the limiting reagent in this experiment? -Nitric acid -Glacial acetic acid -Phenacetin…Thank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 Isobutalene
- Using 1-butyne, CH3CH2CH≡CH, write reaction steps to show how in the laboratory youmay prepare the following compounds. (No reaction mechanism is required)i) 1,1,2,2-Tetra chlorobutane (CH3CH2C(Cl2)CH(Cl2) ii) Butanal (butylaldehyde, CH3CH2CH2CHO)using any necessary reactants and reagents, propose reasonable syntheses of the following compounds from cyclohexanone can you please help with coumpound 3 (I attached it below). Thank you!Devise a synthesis of each of the following compounds. Besidesinorganic reagents, you may use hydrocarbons and halides having ≤ 6C′s, and CH2=CHCOOCH3 as starting materials. Each synthesis must use at least one of the carbon–carbon bond-forming reactions.



