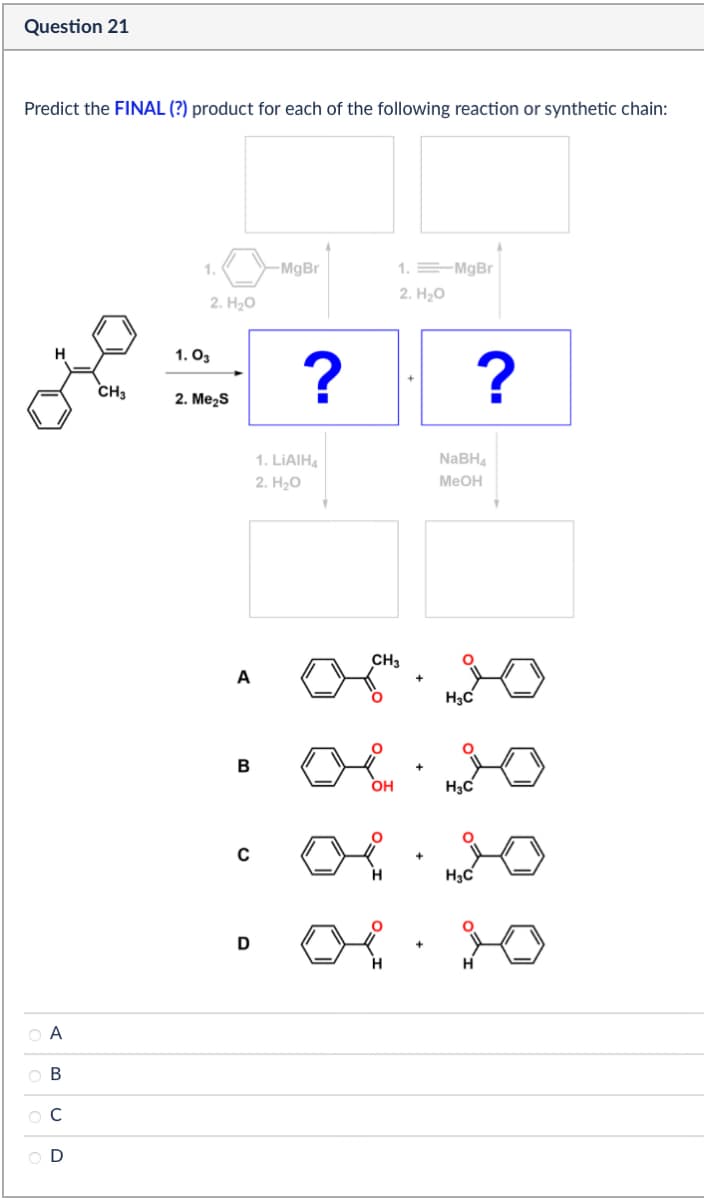
Question 21 Predict the FINAL (?) product for each of the following reaction or synthetic chain: H CH3 ABCD 1. 1.03 2. H₂O 2. Me₂S -MgBr ? 1. MgBr 2. H₂O ? 1. LiAlH4 2. H₂O NaBH4 MeOH A B с D CH3 + H3C 06.20 OH + H3C 06.20 H H3C H
Q: Provide a synthesis of the target molecule. All of the carbon atoms in the final product must come…
A: Step 1: Wittig reaction:Methyl bromide with triphenyl phosphine and n-BuLi gives phosphorus…
Q: Give the mechanism for the reaction - draw it out
A:
Q: Draw the product of the reaction shown < below. Use a dash or wedge bond to indicate the…
A:
Q: Promethium (Z = 61) is essentially nonexistent in nature as all of its isotopes are radioactive.…
A:
Q: Identify the reagent(s) needed for the following reaction to occur: Check all that apply. ▸ View…
A: Step 1: Step 2:Step 3: Step 4:
Q: 2. For a 5.00 g sample of dinitrogen tetroxide (N205), calculate the following: a. The number of…
A: Step 1:The molar mass of N2O5(Dinitrogen pentaoxide) is :2×14.01 g/mol (for Nitrogen)+5×16.00 g/mol…
Q: Huhuhuhuhuu
A: C) The ring with methyl group is activated one because the CH3 group donated electron density inside…
Q: Show the retrosynthetic analysis and foreward reaction of the following compounds. Please draw out…
A:
Q: Radium-226 is radioactive and has a half life of 1600. years. How much of a 1.90 mg sample would be…
A: Given: t1/2=1600yrs;t=1.34yrs;N0=1.90mg;N=???mgStep 1: Write the integrated rate law for first…
Q: Which molecule(s) below is(are) soluble in water? CI H-C-OH CIC CI A Both A and B Neither A nor B B…
A: The question is asking us to determine which of the given molecules are soluble in water. Solubility…
Q: When the Ag* concentration is 1.21 M, the observed cell potential at 298K for an electrochemical…
A: The objective of the question is to find the concentration of Cr3+ in an electrochemical cell…
Q: A weather balloon with a volume of 151 L is launched at 20.0\deg C at sea level, where the…
A: We'll use the ideal gas law, which states: PV=nRT Where:P is the pressure of the gas (in Pascals or…
Q: Show the retrosynthetic analysis and foreward reaction of the following compounds. Please draw out…
A:
Q: None
A:
Q: One of the nuclides in spent nuclear fuel is U-235 an alpha emitter with a half-life of 703 million…
A: The objective of this question is to determine the time it will take for the amount of U-235 to…
Q: a. b. C. CH,CH-CH-CH-CH₂OH CH,CHCH,CHCH,CH3 CH₂CHCH CHCH CH₂ CH3 OH OH Br d. OH CH,CHCH2CH3 OH f.…
A: (a) 2-Pentanol:'Pent' indicates a five-carbon chain.'anol' indicates the presence of an alcohol…
Q: The equilibrium constant, Kp. for the following reaction is 55.6 at 698 K: H2(g) +12(8) 2HI(g)…
A:
Q: A chemist fills a reaction vessel with 6.88 atm hydrogen (H2) gas, 7.31 atm oxygen (O2) gas, and…
A: Given: ΔfG°,kJ/molP,atm2H2(g)0.006.88+O2(g)0.007.31⇌2H2O(g)−228.61.19Step 1: Write the…
Q: Draw the following a) N-methylpropanamideb) cis-2-heptenec) 2-hexanone d) butylbutanoatee)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Please answer in tipping format
A: F)The reaction of CH3-CH2-CO-OH (propionic acid) with PCl3 and 2(CH2)2NH (diethylamine) typically…
Q: 6.66 x 1024 CH4 molecules mol
A: We are given with molecules of CH4 whose number is 6.66 × 1024 molecules.Number of moles can be…
Q: Micelles are colloidal systems that are made of surfactants. Discuss 3 pharmaceutical applications…
A: Let's delve deeper into why micelles are valuable in pharmaceutical applications:1. Surpassing…
Q: For each of the following reactions, please fill in the blanks. CH3 CH3 1. BH3 Et₂O ? 2. H₂O2 H3C-…
A: 1) In hydroboration-oxidation of alkenes, an alcohol is produced in an anti-Markovnikov fashion…
Q: What type of step is this? Check all that apply. Draw the products of the step on the right-hand…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw the product please and explain
A: ### Step 1: Understanding the Basic Steroid StructureSteroids are characterized by a carbon skeleton…
Q: Starting from methane and butane make the following diene. You must use either methane and/or butane…
A: Step 1: In the first step, the butane is converted into 1,3-butadiene by dehydrogenation in the…
Q: Solveeeeeeee
A: Step 1: A) The structure is a phenol derivative with additional alkyl groups attached to the benzene…
Q: Please don't provide handwritten solution...
A: To indicate atoms that are behind or in front of the plane of the paper, use dashed lines and…
Q: Can you please set up the ice table
A: Step 1:Introduction to datas :Given ,Ka for HNO2= 6.8∗10−4Molarity of HNO2= 0.55 MMolarity of…
Q: From the table below, calculate : 1. The order of A 11. The order of B 111. The Rate Law 1. The Rate…
A:
Q: Can you explain the problem? What if it was a base?
A:
Q: Learning AA E4 CH 16 17 and 18 prod03-cnow-owl.cengagenow.com Learning Learning Learning…
A: Barium hydroxide (Ba(OH)2) is a strong base, meaning it dissociates completely in water. We can…
Q: 2. Starting with 2-methyl-1-phenylpropan-1-one, show how you can convert this to 2-methyl-1-…
A: The objective of the question is to convert 2-methyl-1-phenylpropan-1-one to…
Q: Question 15 Choose the correct chemical structure for every single acronym used below: n-BuLi ? A B…
A: Step 1:The acronym "n-BuLi" stands for "normal butyllithium." "n-" Prefix: In this context, "n-"…
Q: Which of the following pairs of structures is not two conformations of the same molecule. H. Mecl H…
A: Conformations are any of the limitless number of probable spatial arrangements of atoms in a…
Q: None
A: The formulas you provided are a mix of calculations and data points:a) 21.8 / 4.79 This is a simple…
Q: A titration was performed using a 25.00 mL sample of 0.15 M weak acid and 0.092 M strong base. What…
A: In this problem, the point of titration is after the equivalence point. Here are some notes to…
Q: O Chemical Reactions Using molarity to find solute moles and solution volume 0/5 Bish A chemist adds…
A: To find the millimoles of barium chlorate (Ba(ClO3)2) added, we can follow these steps:Convert the…
Q: Identify the most stable conformation of trans-1-methyl-4-bromocyclohexane (5). A B Me (5) Me Br A…
A: Step 1:
Q: Identify how you would make pentylamine from 1-hexanol: OH NH2 The transformation above can be…
A: 1-hexanol is first treated with PBr3 to replace -OH with -Br via SN2 reaction.Afterward, a strong…
Q: Complete and balance the equations according to acid and base behavior in water. Phases are…
A: The balanced equation can be shown as:Nitric acid is a strong acid. Therefore it dissociates…
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: →> CH3CH₂OH HCl n + 0 OH CH CH OH 3 HCI
A: Step 1:
Q: 2:15 AM Sun Apr 14 Question 5 of 8 When copper is heated with an excess of sulfur, copper (I)…
A: Given: molCu=0.0970mol;Actualyield,AY=massCu2S=5.36gMCu2S=159.16g/molStep 1: Write the balanced…
Q: A weak base has a K₁ of 5.6x104, determine the pK₁ of the base. 3.25 7.64 10.75 6.36
A: The pKb of a base is the negative logarithmic of Kb value of the base. pKb = - log Kb As per the…
Q: Below is the structure of the drug Odevixebat, which is used for treating decreased bile production.…
A: Step 1:Step 2:
Q: None
A: The provided information has a good starting point, but needs some completion! Let's fix the…
Q: Please don't provide handwritten solution.
A: I'll assume the reaction:Ni(s)+PdCl2(aq)→NiCl2(aq)+Pd(s)At the cathode, reduction occurs.…
Q: An unidentified covalent molecular compound contains only carbon, hydrogen, and oxygen. When 5.60 mg…
A: Step 1:Let formula of the compound is CxHyOzGeneral combustion reaction isCxHyOz + (x + y/4 - z/2)…
Q: How many hydrogens will be attached to the carbons labeled 1, 2, and 3 in the condensed structural…
A: The number of hydrogen atoms attached to each carbon in the condensed structural formula for…
Step by step
Solved in 2 steps with 1 images

- Give the main organic product of the reaction in Figure 6. [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] * Your answerPls solve this question correctly in 5 min i will give u like for sure Balance the chemical equation of a reaction formed by the bromination of trans-cinnamic acid if we started with 0.3088 grams of trans cinnamic acid and 1mL of bromine. The product of this reaction is 2,3 - dibromo - 3- phenylpropanoic acidAcetylene is a very weak acid; however, it will react with moist silver(I) oxide and form water and a compound composed of silver and carbon. Addition of a solution of HCl to a 0.2352-g sample of the compound of silver and carbon produced acetylene and 0.2822 g of AgCl. (a) What is the empirical formula of the compound of silver and carbon? (b) The production of acetylene on addition of HCl to the compound of silver and carbon suggests that the carbon is present as the acetylide ion, C22-. Write the formula of the compound showing the acetyl ide ion.
- Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2OState the reaction happening in each step of the synthesis sequence shown below. CH3CH2OH➡️H2C=CH2➡️CH3CH3➡️CH3CH2Br For the reactants shown below, Name the primary product(s). Show the structure of the product(s).Calculate the % yield-show work in a clear organized matter-be aware of significant figures. Which is the limiting reagent? A Freidel – Crafts Alkylation Name_______________ Data: Alkyl chloride used________tert-butyl chloride___________________________________ Mass alkyl chloride____15.7549 g_______________________________________ Mass 1,4-dimethoxybenzene_____5.0035 g_______________________________ Mass product isolated____2.7834 g______________________________________ Appearance of product__________dry snow white solid________________________________ m.p. rextal product__101 deg Celsius ____________________________________________ Published m.p. product____102-104 o C_____________ source____Chemspider________________ Name of product__1,4-di-tert-butyl-2,5-dimethoxybenzene._______________________________________________
- Make a table of the physical properties (molar mass, MPT, BPT density and mass used/needed) for the reduction of benzophenone lab reactions. (bezhydrol is the product of reaction). Plz do fast asap, urgent.Only need solution of 3rd part. Ans: 10 isomers (Please send their structures)For 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is used
- What is the final organic product of the reaction scheme in Figure 12? [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] *Compounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reaction16.0g of O2 reacts with an excess amount of C3H6. Calculate the ads of H2O produced. C3H8 + 5 O2 -> 4 H2O + 3 CO2

