Q: Defermine the following for a 0.9%m/v of saline solution at 25°C. The density of the solution is 1.0...
A: The freezing point elevation, boiling point elevation and osmotic pressure of a 0.9 % m/v of a salin...
Q: A sample of ammonia goes through a temperature change of +39.63 °C while absorbing 603.9 joules of h...
A: Given, Temperature change (∆T) = 39.63 °C Amount of heat absorbed (Q) = 603.9 J Specific heat capaci...
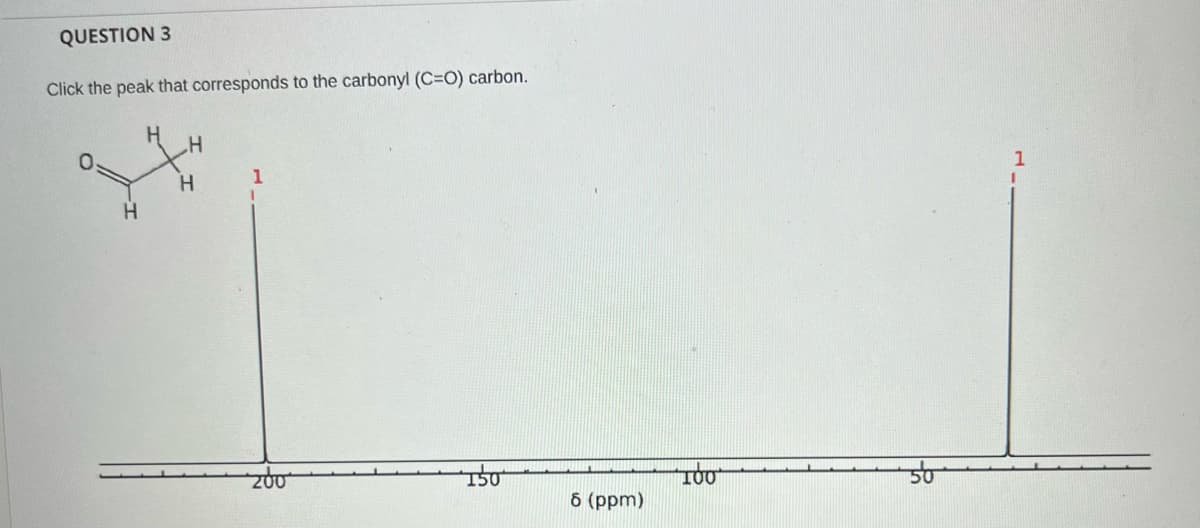
Q: Identify the expected product of the following Cope rearrangement. он OH -OH IV V OI O II OIV
A: It is cope reaction, a type of [3,3]-sigmatropic reaction. I have shown the complete reaction and ...
Q: If the concentration of NO(g) was decreased, 3a) which way would the equilibrium shift? a) to the r...
A: If the reaction is at equilibrium and reversible, then equilibrium is subjected to change in Concent...
Q: A 2.0 L solution contains 1.0 mol of sodium formate (NaCHO,). What is the pH of this solution after ...
A: Henderson - Hasselbach equation is used to calculate the pH of buffer solution . pH = pKa + log[Con...
Q: between CO2, water temperature, pH, and 02 evels. The emphasis should be on how they are all connect...
A:
Q: 0.356=44.032.0x+88.046.0(0.220−x) This question is for chemistry. I'm not sure how to solv...
A: In this question, we have to solve the given equation to find the value of x. 0.356=44.032.0x+88.046...
Q: Draw two amino acids based on serine
A:
Q: Prelaboratory Assignment: Qualitative Analysis Group III Cations 1. Prepare a complete flow chart fo...
A: Group 3 cation involve Cr3+ , Al3+ , Ni2+ and Fe3+.
Q: Draw curved arrows to show electron reorganization for the reaction step below.
A: Given structure of reactant and product
Q: molar solubility of
A:
Q: How many calories would be required to melt 45g of water?
A:
Q: topic: stoichiometry Convert the following from grams to moles of the following. Write your solutio...
A:
Q: Planning A Grignard Synthesis: (1) Identify the Degree of the Alcohol (2) Identify the Carbonyl Func...
A:
Q: The comma between PhO, (8) and PHSO, (s) denotes a heterogeneous mixture of the two solids. The righ...
A:
Q: e major organic product
A:
Q: What mass of H2SO4 would be required to prepare 750. mL of a 0.150 M H2SO4 solution?
A: Given, Molarity of H2SO4 solution = 0.150 M = 0.150 mol/L Volume of H2SO4 solution = 750. mL = 0.750...
Q: formation?
A:
Q: Problem 3: Consider the following buffer solution: 600.0 ml 0.550 M CH,COOH 0.325 M NACH,COO K,1.76 ...
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts fo...
Q: Provide the major organic product(s) that complete the following reaction equations. Please include ...
A:
Q: 2. Given the reactions: Reaction Kc at 25°C 6.8 x 104 HF(aq) H*(aq) + F'(aq) H2C2O4(aq) = 2H*(aq) + ...
A:
Q: Aluminum trifluoride (AIF3) is sparingly soluble with a Ksp value of 1.1 x 10-3. Calculate the molar...
A:
Q: Unknown Analysis: Calculate the mass spec. of 88g per mole. (show calculations and formulas) Below i...
A:
Q: anisms 4) from the compounds in group A and 1 compatible reactant from the compounds in group B that...
A:
Q: 25 nm, which çan be used for the spectrophotometric determination of ammonia. OCF 2 OH + NH3 indophe...
A: Solution -
Q: Present the 3-membered ring pathway for the following reactions. Cl2 Br
A: Organic compounds can be defined as the compounds containing hydrogen and carbon atoms.
Q: Methyl bromide is widely used as a fumigant to prevent the spread of diseases and pests in agricultu...
A:
Q: What happens to your muscles when they get cold? Does that have implications for adding previously f...
A: Given:- What happens to your muscles when they get cold?Does that have implications for adding previ...
Q: How many liters of NH3 can be produced at 60 atm and 24 degrees Celsius from a 80 g sample of nitrog...
A:
Q: Enter your answer in the provided box. The first-order reaction of decomposition of azomethane is gi...
A: Given, Decomposition of azomethane is given below, CH3-N=N-CH3(g) ➝ N2(g) + C2H6(g) This is the firs...
Q: hilic addition reaction with HCI to produce the corresponding tertiary alkyl
A: Organic reactions are those in which organic reactant react to form organic products.
Q: A certain first-order reaction A B is 25% complete in 42 min at 25°C. What is the half- life of the ...
A: Given: The time required for the completion of 25% of a first-order reaction = 42 min We have to cal...
Q: Determine the molar solubility of silver bromide in a. Water b. 6.0 x 10-2 M silver nitrate solution...
A:
Q: Draw reaction mechanism for week 2 A wittig reaction experiment: Week 1 -》 preparation of phospho...
A:
Q: I have an unknown volume of gas at a pressure of 730 mm Hg and a temperature of 315 K. If I raise t...
A:
Q: Describe the appearance and color if: A. Iodine solution is combined with carbon tetrachloride B. ...
A: Please find your solution below : Iodine and its compounds are widely used in analytical chemistry. ...
Q: A solution of acetic acid, HC2H3O2, on a laboratory shelf was of undetermined concentration. If the ...
A:
Q: A blood sample contains 9.2 mM of glucose. If the molar mass of glucose is 180 g/mol, what is the bl...
A: Molarity of glucose = 9.2 mM molar mass of glucose = 180 g/mol
Q: 76 Consider Reactions A, B, and C with activation energies, E, of: 35; 45; and 55 kJ/mol, respective...
A: 76 ) Arrhenius equation is given by, k = A.e-Ea/RT k = rate constant A = Arrhenius co...
Q: Arrange the following ions based on their order of stability.
A: Order of stability of organic carbocation can be explained using resonance and aromaticity as follow...
Q: H3CH2CO¯ Na* ethanol Br
A: Base can abstract most acidic hydrogen at beta position and form alkene.
Q: What are the accompanying changes of: A. Iron rusting B. Burning wood C. Cooking an egg D. The expl...
A: There are two types of changes in the universe- physical change and chemical change.
Q: what are the grams of excess reactant, A reaction mixture initially contains "22.35" g Fe2O3 and 14....
A: Given reaction: Fe2O3(s)+3CO(g)→2Fe(s)+3CO2(g) The reaction mixture initially contains 22.35 g of Fe...
Q: For the hypothetical reaction A+ 38- 2C, the rate should be expressed as Mutiple Choice rate (VINABY...
A: Given :- A + 3B → 2C To write :- expression for rate of reaction in terms of change in concentrati...
Q: Stomach acids (HCL) has a pH of about 1.5. For individuals who suffer from acid refulx, antacids or ...
A: We have to predict the balance chemical reaction.
Q: Give the description of accompanying changes of: A. Iron rusting B. Burning wood C. Cooking an ...
A: a. Iron rusting Answer - According to the question - Rusting of iron is a chemical change because a ...
Q: O STATES OF MATTER Interconverting molar mass and density of ideal... A sample of an unknown compoun...
A:
Q: Given the dissolution of silver phosphate: A93PO4 3 Ag* + PO43- Which of the following conditions wi...
A: The equilibrium reaction given is Ag3PO4 ⇌ 3 Ag+ + PO43– ----------- (1)
Q: (b) mass spectroscopy. Explain any one of them. Mention different ionization modes in
A:
Q: The_Li²* ion is hydrogenic and has a Lyman series at 740,747 cm1, 877,924 cm1, 925,933 cm, and beyon...
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Please help me solve the following question and explain it, I cant seem to get the right answer for some reason. make sure its correct 1000% thank you sm!!I need a confirmation with a hundred percent confidence on my answer a lot of controversy on the correct answer so I will just take your word for it so please be surepls complete the 2nd table given the data1. 500.0 mL 0.2 M KI2. 500.0 mL 0.2 M KCl3. 500.0 mL 0.1 M K2S2O84. 500.0 mL 0.1 M K2SO45. 500.0 mL 4.0 mM Na2S2O3 (from Na2S2O3∙5H2O)
- Identify what separation technique can be performed to do the following. ANSWER ONLY 1-10Subject :- Chemistry how do I convert if I have 112 mg/L as CaCO3 and they are asking me in meq/L?show the complete solution and write answers in 3 significant figures. DYE CALCULATED Rf Tartrazine (yellow #5) ? Sunset yellow (yellow #6) ? Allura red (red #40) ? Brilliant blue (blue #1) ? M&M's color COMPOSITION Orange ? Green ? Blue ? Brown ? CHROMATOGRAM BEFORE: (image 1) CHROMATOGRAM AFTER: (image 2)

