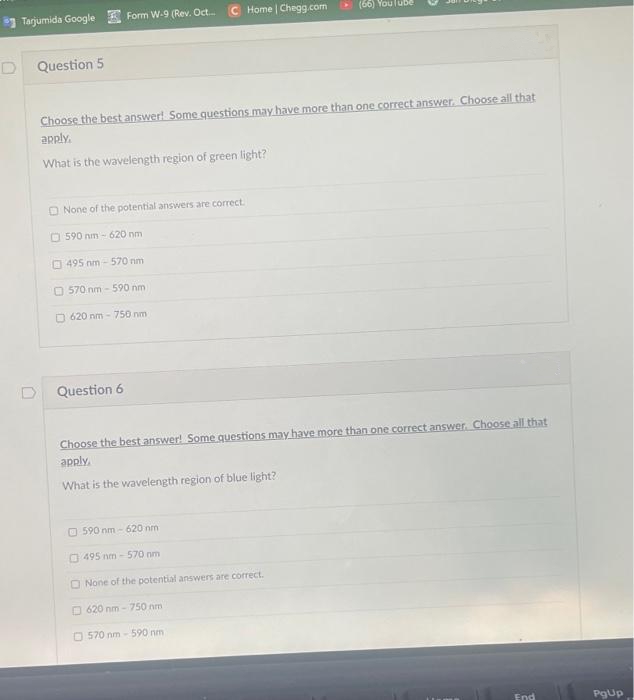
Question 5 Choose the best answer! Some questions may have more than one correct answer. Choose all that apply. What is the wavelength region of green light? O None of the potential answers are correct. O 590 nm - 620 nm O 495 nm - 570 nm O 570 nm - 590 nm O 620 nm - 750 nm D Question 6 Choose the best answer! Some questions may have more than one correct.answer. Choose all that apply. What is the wavelength region of blue light? O 590 nm - 620 nm O 495 nm - 570 nm O None of the potential answers are correct. O 620 nm - 750 nm O 570 nm - 590 nm
Question 5 Choose the best answer! Some questions may have more than one correct answer. Choose all that apply. What is the wavelength region of green light? O None of the potential answers are correct. O 590 nm - 620 nm O 495 nm - 570 nm O 570 nm - 590 nm O 620 nm - 750 nm D Question 6 Choose the best answer! Some questions may have more than one correct.answer. Choose all that apply. What is the wavelength region of blue light? O 590 nm - 620 nm O 495 nm - 570 nm O None of the potential answers are correct. O 620 nm - 750 nm O 570 nm - 590 nm
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter4: Introduction To Quantum Mechanics
Section: Chapter Questions
Problem 17P: In a FranckHertz experiment on sodium atoms, the first excitation threshold occurs at 2.103 eV....
Related questions
Question
5
Transcribed Image Text:*1 Tarjumida Google Form W-9 (Rev. Oct.
Home | Chegg.com
(66) YouluBe
Question 5
Choose the best answer! Some questions may have more than one correct answer. Choose all that
apply.
What is the wavelength region of green light?
O None of the potential answers are correct.
O 590 nm- 620 nm
O 495 nm - 570 nm
O 570 nm - 590 nm
O 620 nm - 750 nm
Question 6
Choose the best answer! Some questions may have more than one correct answer. Choose all that
apply
What is the wavelength region of blue light?
O 590 nm - 620 nm
O 495 nm - 570 nm
O None of the potential answers are correct.
O 620 nm - 750 nm
O 570 nm - 590 nm
End
PgUp
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning