QUESTION 7 Waking fast can consme 5Dkeal perminte Hoer many hours of eecbe are reqired to consume 450 kcal, the energy in a large candy bar? OA 125 O8175he OC75 O015 OE1 QUESTION S Which of the looing does not have a untorm composition troughout OA Cempound OB Heterogeneous mature OC Hamogeneous mdure OD Element OE Solvent Click Sove ond Submit to paer and submit. Click Sene Ansors o sane l aners.
QUESTION 7 Waking fast can consme 5Dkeal perminte Hoer many hours of eecbe are reqired to consume 450 kcal, the energy in a large candy bar? OA 125 O8175he OC75 O015 OE1 QUESTION S Which of the looing does not have a untorm composition troughout OA Cempound OB Heterogeneous mature OC Hamogeneous mdure OD Element OE Solvent Click Sove ond Submit to paer and submit. Click Sene Ansors o sane l aners.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 63QAP: Butane gas, C4H10, is sold to campers as bottled fuel. Its density at 25C and 1.00 atm is 2.38 g/L....
Related questions
Question
100%
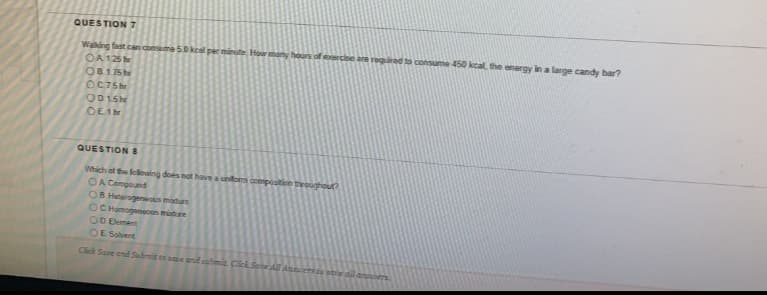
Transcribed Image Text:QUESTION 7
Waking fast can consume 50keal per minute Hor many hours of eercbe are required to consume 450 kcal, the energy in a large candy bar?
OA 125
O8175he
OC75
OD15
OE1
QUESTION 8
Which of the lellwing does not have a unom composition troughout?
OA Compound
OB Heterogeneous mature
OC Homogeneous midure
OD Element
OE Solvent
Click Save ond Sumit to save and submit. Click Sene All Ansers to pae dil anrs
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning