Question 76 of 96 How many iters of a 0.209 M KI solution is needed to completely react with 2.43 g of Cu(NO,) according to the balanced chemical reaction 2 CUNO,lag) + KI(aq) -- 2 Cultag) + (s) + KNO,(aq) molCuNo 0.0310 LKI 243 0.124 LKI 187.57 ACupo molCuNO 0.209 2 STARTING AMOUNT 1 mol Cu(NO) 4 mol KI 0.0310 L KI 243g CuNO,), * - 0.124 L KI 187.57 e CuNO,) 2 mol Cu(NO,) 0.209 mol KI AO FACTOR DELETE ESET •( ) 0.0310 2.43 187.57 0.209 0.00542 6.022 x 10 0.124 0.0620 2 166 o CuNO,) LKI mL Cu(NO,). mol Cu(NO,), MKI mol KI mL KI LCu(NO,
Question 76 of 96 How many iters of a 0.209 M KI solution is needed to completely react with 2.43 g of Cu(NO,) according to the balanced chemical reaction 2 CUNO,lag) + KI(aq) -- 2 Cultag) + (s) + KNO,(aq) molCuNo 0.0310 LKI 243 0.124 LKI 187.57 ACupo molCuNO 0.209 2 STARTING AMOUNT 1 mol Cu(NO) 4 mol KI 0.0310 L KI 243g CuNO,), * - 0.124 L KI 187.57 e CuNO,) 2 mol Cu(NO,) 0.209 mol KI AO FACTOR DELETE ESET •( ) 0.0310 2.43 187.57 0.209 0.00542 6.022 x 10 0.124 0.0620 2 166 o CuNO,) LKI mL Cu(NO,). mol Cu(NO,), MKI mol KI mL KI LCu(NO,
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter15: Solutions
Section: Chapter Questions
Problem 95AP: 95. Many metal ions form insoluble sulfide compounds when a solution of the metal ion is treated...
Related questions
Question
Help me, please. Please write the labels with number each conversion factor.
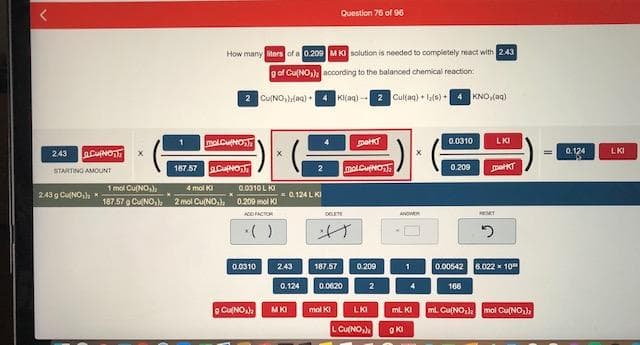
Transcribed Image Text:Question 76 of 96
How many iters of a 0.209 M KI solution is needed to completely react with 2.43
g of Cu(NO,) according to the balanced chemical reaction
2 CUNO,lag) +
KI(aq) -- 2 Cultag) + (s) +
KNO,(aq)
molCuNo
0.0310
LKI
243
0.124
LKI
187.57 ACupo
molCuNO
0.209
2
STARTING AMOUNT
1 mol Cu(NO)
4 mol KI
0.0310 L KI
243g CuNO,), *
- 0.124 L KI
187.57 e CuNO,) 2 mol Cu(NO,) 0.209 mol KI
AO FACTOR
DELETE
ESET
•( )
0.0310
2.43
187.57
0.209
0.00542 6.022 x 10
0.124
0.0620
2
166
o CuNO,)
LKI
mL Cu(NO,). mol Cu(NO,),
MKI
mol KI
mL KI
LCu(NO,
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning