Question: A solution is to be prepared in a laboratory. The solution requires 0.0465 mol of quinine (C20H24N2O2). What mass, in grams, should the laboratory technician obtain in order to make the solution? Below is a picture of how all the work should correctly look in this Dimensional Analysis format:
Question: A solution is to be prepared in a laboratory. The solution requires 0.0465 mol of quinine (C20H24N2O2). What mass, in grams, should the laboratory technician obtain in order to make the solution? Below is a picture of how all the work should correctly look in this Dimensional Analysis format:
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter3: Measurement And Chemical Calculations
Section: Chapter Questions
Problem 43E: a 5.74 cg to g, b 1.41 kg to g, c 4.54 x 108 cg to mg
Related questions
Question
Please answer this question correctly.
Question: A solution is to be prepared in a laboratory. The solution requires 0.0465 mol of quinine (C20H24N2O2). What mass, in grams, should the laboratory technician obtain in order to make the solution?
Below is a picture of how all the work should correctly look in this Dimensional Analysis format:
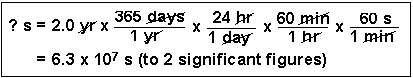
Transcribed Image Text:24 hr , 60 min
60 s
365 days
1 yr
? s = 2.0 yr x
1 day
1 br
1 min
= 6.3 x 107 s (to 2 significant figures)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

