Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter9: Nucleophilic Substitution And Β-elimination
Section: Chapter Questions
Problem 9.58P: Using your reaction roadmap as a guide, show how to convert cyclohexane into hexanedial. Show all...
Related questions
Question
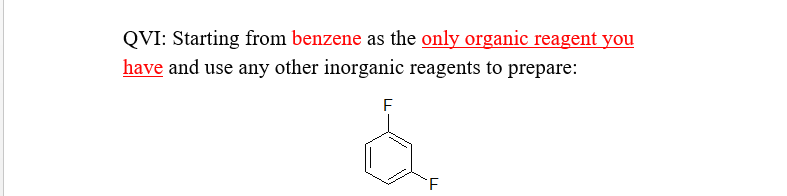
Transcribed Image Text:QVI: Starting from benzene as the only organic reagent you
have and use any other inorganic reagents to prepare:
Expert Solution
Step 1
Starting from benzene, nitration would yield nitrobenzene and further reaction with xenon fluoride in DCM, reduction of nitro group changes to amine and fluorine is substituted at meta-position. This undergoes a diazonium reaction followed by treatment with HBF4 and heat (Scheizmann reaction) to give the desired product.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
