Q: Consider 1-bromopropane, CH3CH2CH2Br. (a) Which of these is the lowest energy conformation?
A: The newman projection of 1 bromopropane shows the lowest energy conformation is Anti staggered form…
Q: Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which …
A: (a)
Q: Which of the following chair structures represents the most stable conformation of the…
A: If the substitutents are at 1 and 4 positions and cis to each other in cyclohexane, then the most…
Q: Arrange the following sets of compounds based on the property given in increasing order. Type in the…
A: The carbocations given are,
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to…
A: The isomers of C6H14 are as follows: A. Consider the treatment of 2-methylpentane with Cl2 + hv.…
Q: Haloalkane with molecular formula, C,H,CI has four structural isomers, A, B, C and D. The structure…
A: Let us write the IUPAC names of A,B,C
Q: Arrange the compounds 2-methyl-1-butene, 2-methyl-2-butene, and 3-methyl-1-butene in order of…
A: The given compounds (2-methyl-1-butene, 2-methyl-2-butene, and 3-methyl-1-butene) have to be…
Q: Predict the relative stabilities of alkenes and cycloalkenes based on their structure and…
A: SOLUTION: Step 1: more substituted alkenes are going to be more stable compared to less substituted…
Q: Which do you expect to be the more stable conformation of cis-1,3-dimethylcyclobutane, A or B? Why?
A: Conformers are any two arrangements of atoms in a molecule that differ by rotation about single…
Q: 1.1 Build a model for line structure and use the template given to draw a Newman projection for this…
A: The Newman projection is a way of drawing different conformations that are possible along the C-C…
Q: 6. a. Draw the following molecule as a flat ring skeletal structure with correct stereochemistry b.…
A: Cyclohexane has different conformations such as chair form, boat form, twist boat and half chair.…
Q: Which of the following statements about cyclooctatetraene (the Lewis structure of which could be…
A:
Q: 7. Which one of the following conformations of disubstituted cyclohexanes is expected to be the most…
A: Presence of one bulky methyl group at axial position will cause two gauche butane interaction. This…
Q: 5. Which of compounds is likely to adopt a planar conformation? Why? .. A. С. D. В.
A: A. Cycloheptatriene cation .Here all the carbons are Sp2 hybridised and contain 6 pi electrons .so…
Q: a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed.…
A: Hydroboration is a syn addition. Hydrogen and boron add across the double bond on the similar side…
Q: Given the molecular formula, calculate the degree of unsaturation compounds. Suggest at least three…
A:
Q: How many rings and π(pi) bonds are contained in compound A and draw one possible structure for this…
A:
Q: Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider…
A: The structure(s) of all of the alkene isomers, C5H10, that contain a branched chain with E/Z…
Q: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
A:
Q: Draw the possible constitutional isomers having molecular formula C8H16 that meet all of the…
A: In order to know constitution isomers we need to determine the degrees of unsaturation:number of pi…
Q: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
A:
Q: Nhen compound X undergoes ring-flipping, which compound below represents its ring-flipped conformer?…
A: We have given the organic compound i.e. 1,2diethyl-3-methyl cyclohexane ring we have to gives its…
Q: The energy difference between cis- and trans-but-2-ene is about 4 kJ/mol; however, the trans isomer…
A: Given molecule: cis- and trans-but-2-ene 4,4-dimethylpent2-ene
Q: Predict the more stable bridge head alkene in the following compounds. (A) (В) (C) (D)
A: According to Bredt's rule a double bond cannot be placed at the bridgehead of a bridged ring system…
Q: Draw all possible stereoisomers of 1,3-dimethylcyclopentane and give the relationships between them
A:
Q: Using what you know about the conformational energetics of substituted cyclohexanes, predict which…
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: Conceptual: Rank the following alkenes in decreasing order of stability. b
A: The relative stability of the alkenes can be understood by the presence of hyperconjugative H atoms.…
Q: Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to…
A: The isomers of C6H14 are as follows:
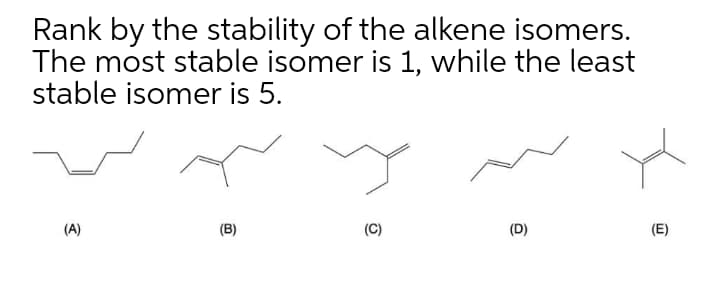
Q: Rank the stability of the following alkenes from least to most stable. а. b. с. (А) а, b, с (B) b,…
A:
Q: Which pair of structures are constitutional isomers? (a) A and B (b) A and C (c) C and D (d) all are…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: Draw the structure consistent with each description. a.(2E,4E)-octa-2,4-diene in the s-trans…
A:
Q: Draw a structural formula for the cycloalkene with the molecular formula C6H10 that reacts with Cl2…
A: An addition reaction is a reaction in which the addition of molecules to the unsaturated molecules…
Q: Treatment of cyclohexene with C6H5CHI2 and Zn(Cu) forms two stereoisomers of molecular formula…
A: The given reaction is an example of a Simon-smith reaction in which the reaction of an alkene with…
Q: CH3 CH3 CH3 CH3 Compound I Compound II (i) Are the above cyclohexane derivatives cis- or…
A:
Q: 2. Draw the most stable conformation of the following: (a) trans-1-butyl-3-methylcyclohexane (b)…
A: Bulky group should be present at equitorial position for stability so that it can feel minimum…
Q: A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having…
A: The molecular formula of the compound is C6H10. The compound contains 6 carbon atoms and 10 hydrogen…
Q: Measure the C-C-C and C-C-H bond angles in the energy-minimized model of cyclohexene and compare…
A: In the energy-minimized model of cyclohexene, for C-C-C and C-C-H bonds, the possible bond angles…
Q: What are the relative energy levels of the three staggered conformations of 2,3-dimethylbutane when…
A: Given: 3 Different conformation and some of the statements regarding it. To find: The correct…
Q: (4B) Carbocation Stability: Rank the following carbocations in order of stability and briefly…
A: Carbocation is species in which carbon bears positive charge.
Q: Write Newman projections for all three staggered conformations of (R)-2-bromobutane, viewing down…
A: (R)-2-bromo butane produces three staggered conformations. A produces trans-2-butene (major). C…
Q: Draw a structural formula for the cycloalkene with the molecular formula C6H10 that reacts with Cl2…
A: In the given product, two chlorine atoms are attached to adjacent carbons and the orientation is…
Q: The anti-conformation is the most stable n-butane conformation. If you could take a snapshot of a…
A: Anti-conformation of n-butane is the most satble conformation. To determine, for 1 mole of n-butane…
Q: Rank the alkenes from most stable to least stable. A B H3C CH3 H3C H H2C=CH2 H3C CH3 H3C The ranking…
A: Hyperconjugation is one of the factors which depend upon stability of alkenes. As the number of…
Q: (S)-1-Bromo-1,2-diphenylethane reacts with the strong base CH3CH2ONA to produce the cis- stilbene…
A: E2 elimination The reaction that involves only one step mechanism is called E2 elimination. The…
Q: Which of the following radicals are allylic? B D. only D A, B, and C B and C
A: The radicals given are,
Q: The diaxial conformation of cis- 1,3-dimethylcyclohexane is approximately 23 kl/mol (5.4 kcal/mol)…
A: We have been asked to draw the chair conformation and show the unstability of diaxial conformation.
Q: Compound X has the formula C35H54BR2. When X is treated with excess H2 over Pd/C, the product has…
A:
Q: 2. How many units of unsaturations does a compound with the formula C25H40Cl4 have? 3. Give the name…
A: For question 2, we have to find the number of units of unsaturation in the compound C25H40Cl4. For…
Q: .41 Draw the more stable chair conformation of (a) (b) (c).
A: A substituted cyclohexane conformation is any of several three-dimensional shapes adopted by a…
Q: 6. In an organic chemistry lab, chemists were attempting to convert compound I into compound III via…
A: The leaving group attached to 3o carbon. Thus, nucleophilic substitution reaction must be occurred…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Draw the structure(s) of all of the alkene isomers, C5H10, that contain a branched chain. Consider E/Z stereochemistry of alkenes.a) When (Z)-3-methylhex-3-ene undergoes hydroboration–oxidation, two isomeric products are formed. Give their structures, and label each asymmetric carbon atom as (R) or (S). What is the relationship between these isomers?Compound A and compound B are in equilibrium. Write a stepwise mechanism from compound Ato compound B showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and products are formed. Show all lone pairs and formal charges. Lastly, explain which compound (Aor B) will be in higher concentration.
- Which will be more stable, cis or trans-1,4-tert-butylcyclohexane? Explain by drawing their structures?A compound C4H11N is known from its reactivity andspectroscopic properties to have no hydrogen atomsattached directly to the nitrogen atom. Write all structuralformulas consistent with this informationWhich of the statement is INCORRECT? a. The increase in stability of 2,4-hexadiene over 1,3-hexadiene is due to the increased double bond substitution of the former. b. The stabilization of dienes by conjugation is less pronounced than the aromatic stabilization of benzene. c. Resonance description in alkenes usually involves charge separation. d. Higher energy pi-orbitals often have decreasing number of nodes.
- Arrange the following compounds in the increasing order of reactivity towards Conc.HNO3 & Conc.H₂SO4 1. Benzene 2. Chlorobenzene 3. phenol 4. Toluene 5. Nitrobenzene A 1.2.3.4.5 (B) 5.1,243 5.1.4.2.3 D 5.2.1.4.3Five isomeric alkanes (A–E) having the molecular formula C6H14 are each treated with Cl2 + hv to give alkyl halides having molecular formulaC6H13Cl. A yields five constitutional isomers. B yields four constitutionalisomers. C yields two constitutional isomers. D yields threeconstitutional isomers, two of which possess stereogenic centers. Eyields three constitutional isomers, only one of which possesses astereogenic center. Identify the structures of A–E.Haloalkane with molecular formula, C,H,CI has four structural isomers, A, B, C and D. The structure of A, B and C are shown below. A - (CH3)3CCI B - CH3(CH2)3CI C - CH3CH(CI)CH₂CH3 Give the IUPAC names and classification for isomers A, B and C, respectively. Arrange isomers A, B and C in ascending order of their reactivity with respect to SN1 reaction. Explain your answer. Write the reaction equations involved for the reaction of the most reactive isomer among A, B and C, with respect to SN1 reaction with aqueous KCN, in ethanol under reflux. Show the reaction mechanism. Draw the structure and give IUPAC name of isomer, D. Isomer D reacts with magnesium in anhydrous ether producing an alkyl magnesium chloride E which is then hydrolyzed and produced F, C4H10. Elimination of D with ethanolic KOH under reflux produces G. The treatment of D with sodium metal in ether solvent yields a symmetry alkane H, C8H₁8, which possesses two tertiary carbons. Hydrolysis of D with aqueous sodium…
- How many rings and π(pi) bonds are contained in compound A and draw one possible structure for this compound A. Compound A has molecular formula C6H10 and is hydrogenated to a compound having molecular formula C6H12Compound A (C6H12O2) reacts with water, acid, and heatto yield compound B (C5H10O2) and compound C (CH4O).Compound B is acidic. Deduce possible structures of compounds A, B, and C1. what are the differences between acyclic and cyclic forms in terms of their physical properties? 2. what is the role of vinegar in liquid-liquid extraction?



