Q: The following reaction in aqueous solution has K = 1 × 1085 at a temperature of 25 °C. 7103(aq) +…
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of…
Q: A student is examining a bacterium under the microscope. The E. coli bacterial cell has a mass of m…
A:
Q: 1. Draw the structure and interaction that occurs between the following R groups of amino acids in…
A: a)The interaction that can occur between the R groups of asparagine and serine is a hydrogen bond.…
Q: Observations: Test Tube A B C D E F Carboxylic Acid acetic acid acetic acid butyric acid benzoic…
A: The reaction between a carboxylic acid and an alcohol in the presence of catalytic amount of mineral…
Q: The image below represents a gaseous equilibrium system for two reactive gases, R and P. The spheres…
A: Given that, an equilibrium reaction system of R → P is shown below Also, given that, the enthalpy…
Q: What is the major organic product or products for this reaction? Select an answer and submit. For…
A: In the above reaction an epoxide ring is treated with CH3CH2MgBr (Grignard reagent) then H3O+. This…
Q: How many moles of sodium hydroxide would have to be added to 225 mL of a 0.349 M nitrous acid…
A: Given data is Volume of nitrous acid solution= 225 mL Molarity of nitrous acid solution= 0.349 M…
Q: Is the Haber process for the industrial synthesis of ammonia spontaneous or nonspontaneous under…
A: Answer: To check the spontaneity of the reaction we have to use below shown relation: ∆G°=∆H°-T∆S°…
Q: Given the Newman projection if the most stable conformational isomer, create the Newman projection…
A:
Q: Find [CO32-] of a 0.070 M carbonic acid solution. Express your answer using two significant figures.…
A:
Q: Study Guide Chapter 16 1. The pl of glycine is 6.0. Draw the condensed structural formulas for…
A:
Q: Amylose is a large polysaccharide composed of glucose units linked with alpha 1,4 linkages. Choose…
A: Reducing sugar and non-reducing sugar are two categories of sugars that differ in their ability to…
Q: How can d8 high spin and low spin in a square planar be drawn?
A:
Q: An aqueous solution at 25 °C has a H₂O* concentration of 5. × 10 M. Calculate the OH concentration.…
A: Answer: In this question concentration of H3O+ ion is given to and we have to find out the…
Q: Click in the answer box to activate the palette. We can use bond-line formulas to represent alkenes…
A:
Q: Where would the compound shown in Image 6 undergo bromination with NBS and benzoyl peroxide?…
A: NBS N bromination succinamide used Allylic bromination is the replacement of a hydrogen on a carbon…
Q: Select the correct product for this reaction Select an answer and submit. For keyboard navigation,…
A: When ketone reacts with grignard reagent followed by hydrolysis gives 3° alcohol.
Q: RECORD ALL DATA IN INK using the correct number of significant figures and correct units. SHOW…
A:
Q: Br C=N
A: The given molecule synthesized in five step with different type of reagent which is shown in below-
Q: Concentrated sulfuric acid is used as a catalyst for the Fischer esterification. Why is sulfuric…
A:
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A: Ba(CHO2)2 is a salt of strong base Ba(OH)2 and weak acid HCHO2 In aqueous solution, Ba(CHO2)2 → Ba+2…
Q: 4. If I add the following will the system shifts forward, backward or remains the same? Ag₂CrO4(s) =…
A: According to the Le Chatelier's principle, "when a stress (change in concentration temperature and…
Q: Studying the reaction A + B → Products, you gather the following information: [A] 0.10 0.30 0.40 [B]…
A:
Q: (25,3R)-1,2-dibromo-3-methylhexane (2R 3R)-1,2-dibromo-3- methylhexane…
A: The explanation had been done below. The concept used here is from General Organic chemistry.
Q: Calcium carbonate is a common ingredient in antacids that reduces the discomfort associated with…
A: Given that : pH of the solution = 1.81 The mass of CaCO3 used for neutralization = 17.0 mg The molar…
Q: In the Friedel-Crafts alkylation of benzene, dialkylation is often a significant by-product. In the…
A: Given that, in the Friedel-Crafts alkylation of benzene, dialkylation is often a significant…
Q: Which of the following best estimates the percentages of the three isomeric methylanilines from the…
A: This question is based on benzyne mechanism.
Q: A cyanide solution with a volume of 13.38 mL was treated with 28.00 mL of Ni²+ solution (containing…
A: Given :- Volume of EDTA required for original Ni2+ solution = 38.84 mL = 0.03884 L Molarity of…
Q: For each of the following chemical reactions with provided rate law, give the overall reaction…
A: The rate law is given for all reaction . We just have to find order of reaction so we will see power…
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A: Answer:- This question is answered by using the concept of determination of nature of the salt . If…
Q: Instructions: DRAW a structure corresponding to each name a. 3-hexanamine b. N-methylpentylamine c.…
A: The given reactions are examples of amines with variable carbon chain and substituents.
Q: What quantity of heat (in kJ) will be released if 1.20 mol of SrO is mixed with 0.627 mol of CO₂ in…
A:
Q: Draw the neutral organic compound that is obtained from the reaction. N 8 1. LIAIH4, ether 2. H30*…
A:
Q: Fill in the blanks. (a) 700 m = km (b) 6.5 cm = mm (c) 610 mL = L (d) 3.4 L = mL (e) 550 g = kg (f)…
A: Fill in the blanks of the following conversions:
Q: Which one of the following substituents is deactivating and ortho-para directing in electrophilic…
A: The question asks which of the four given substituents is deactivating and ortho-para directing in…
Q: NH NO CH3 N CH3 N
A: An aromatic compound is a cyclic compound that exhibits a special type of stability called…
Q: If 205 g of MgCl2 (FW 95.21 g/mol) is added to a solution containing 295 g of (NH4)3PO4 (FW 149.09…
A: To solve this problem we have to find the mass of precipitate formed .
Q: 10. Classify each of the following alcohols as primary (1°), secondary (2°), or tertiary (3°); draw…
A: The question requested is 10. The carbon atom containing alcohol has only one alkyl group is known…
Q: Besides polarimetry, name another method that could be used to assess the enantiomeric excess of a…
A: Enantiomeric excess (ee) is a measure of the purity of a sample of a chiral compound, specifically a…
Q: A student heated a 2.00 g of a hydrate of CoCl2 to drive off the water. After cooling the sample,…
A: Answer: During the crystallization of a compound some water molecules are left in its structure and…
Q: B. Determine the pressure, volume, or temperature of a gas under a certain set of changing…
A: Given, initial volume = 150 mL initial pressure = 760 torr final pressure = 740 torr to…
Q: At 25 °C, the following concentrations were found for the gases in an equilibrium mixture for the…
A: Given that, an equilibrium reaction is N2Og + NO2g ⇌ 3NOg. The equilibrium concentrations are [NO2]…
Q: (1) LIAIH (2) H₂O ОН OH e a r B) C) D)
A: LiAlH4 is very good reducing agent. It can reduce by transferring hydride ion (H-) to electrophilic…
Q: can the following convension performed? Provide the methods acome with the support of mechanism. อ Ⓒ…
A:
Q: Will the following reaction proceed as written? Select an answer and submit. For keyboard…
A: To draw the product of the given organic reaction.
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: ♠ Based on the pH scale,A solution which have pH greater than 7 (pH>7 ) are basic solutions,the…
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A:
Q: A tank contains 60 kg of salt and 1000 L of water. A solution of a concentration 0.03 kg of salt per…
A: Solution - (a) According to the question - Given - Initial concentration = salt / water plug the…
Q: buffer 2: buffer 3: CH3COOH HPO4²- OC H CH3COO H₂PO4 k 3. Write a reaction equation to show how the…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: Consider the reaction of 60.5 mL of 0.310 M NaC7H5O2 with 50.0 mL of 0.245 M HBr. (Ka of HC7HsO2 =…
A: When HBr reacts with NaC7H5O2, NaBr and HC7H5O2 are formed. Now, the Ka value of HC7H5O2 is…
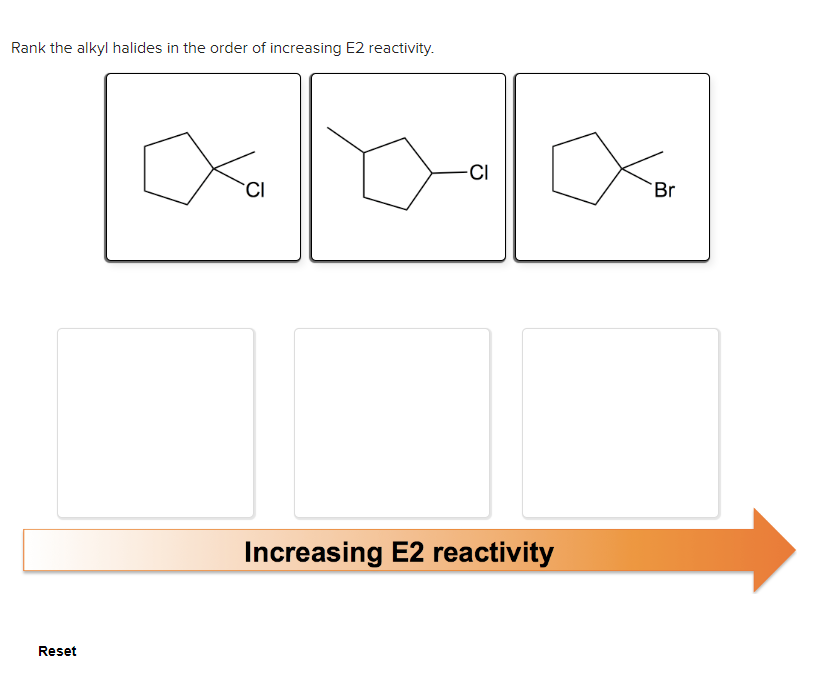
Ochem chapter 7 question 28
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What is the major E2 elimination product formed from each halide?Arrange the alkyl halides in order of increasing reactivity in an SN2 reaction with KI in acetone (least first). I, IV, III, II II, III, I, IV IV, I, III, II III, II, IV, IArrange the following alkyl halides in increasing reactivity in E2 reaction. Write 1 for the least reactive, then 2 and 3 for the most reactive Answer: A. _____ B. _____ C. _____
- Rank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b.−OH, Br−, F− (polar aprotic solvent) c.H2O, −OH, CH3CO2−Which of the following reaction conditions will NOT FAVOR SN1 reaction? a. Sterically hindered alkyl halides b. All of these conditions will favor SN1 c. Reaction with strong base d. Reaction with alcoholsRank the following compounds in order of increasing SN2 reaction rate with a good nucleophile. Explain yourreasoning. (CH3)2CHBr (CH3)3CBr CH3CH2Br CH3CH2I
- Which alkyl halide will react the fastest in an SN2 reaction (CH3)3CI CH3CH2Br CH3CH2I CH3I CH3BrThere are two benzylic bromides that give (Z)-4,6-dimethyl-3-phenyl-3-heptene on E2 elimination. Draw them.What alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict the major product.



