Q: NH он d) O,N- -NH2 -NH2 O,N- -NH2 -NH2 CH3
A: Bases are defined as the molecules which can donate it's electron pair. The more easily a molecule…
Q: Rank the following compounds in the order of INCREASING acidity. но но он II II IV
A: Ans) 2<4<3<1
Q: Rank the nitrogens below in order of decreasing basicity (most basic to least basic) NH2 NH2 .N. EtO…
A: The most basic nitrogen is the one which are more easily available for donation. So resonance effect…
Q: H2 NH2 NH3 -NH2 -NH2 с. O,N CH d. CgH;NH, (CHs)2NH -NH2
A: In case of nitrogenous compound greater the availability of lone pair on nitrogen atom greater it's…
Q: Rank the following compounds in order of increasing basicity in aqueous solution, east basic first…
A:
Q: Rank the following in order of increasing acidity. HS. H3C° НО. A
A:
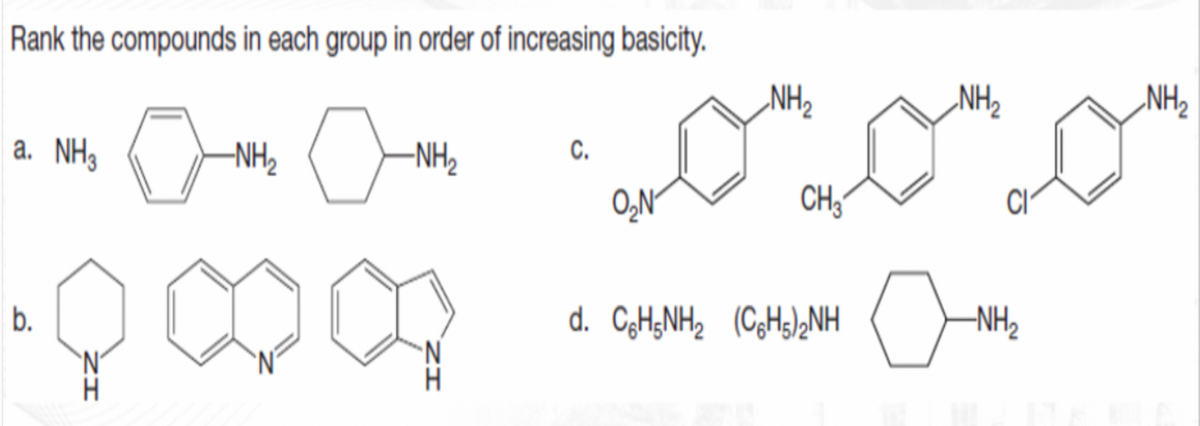
Q: Rank the compounds in each group in order of increasing basicity. NH2 NH2 NH2 a. NH3 -NH2 -NH2 ON CH…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Which of (a)-(d) shows the increasing order of basicity of compounds 1-4? 1 NH₂ O a. 4 <3 <2<1 O b.…
A: Basicity is tendency to accept H+ or we can say availability of lone pair for H+ . Basicity is…
Q: Arrange the following compounds in order of decreasing their acidity (most first) I. H20 II. C2H6…
A: The acidic strength of any compound depends on the stability of their conjugate bases. with the…
Q: NH2 NH2 NH2 NO2
A: The given molecules are: All of these compounds are basic in nature and basicity is inversely…
Q: Rank the labeled protons (Ha–Hc) in mandelic acid, a naturally occurring carboxylic acid in plums…
A: The Hc will the most acidic hydrogen because the conjugate base formed by the deportation of this…
Q: HN-CH3 CH2 NO2 H2C-CI COOH COOH ČOOH II III
A: In this question, we will arrange all these three Compounds in Increasing order of Acidity. You can…
Q: 2-Hydroxybutanedioic acid occurs naturally in apples and other fruits. Rank the labeled protons…
A:
Q: Rank the following compounds in order of their basicity, from most to least basic: NH₂ most least…
A: Given that, three compounds are CH3CH2NH2 CH3CH2O- CH3COO-. We have to rank the given compounds…
Q: bel the three bold hydrogens in compounds A, B, and C in order of acidity, with 1 being the most c…
A: Acidity can be defined as tendency of a compound to lose hydrogen ion.
Q: 9. Final product? SOc B. 10. Put nitrogens in order of basicity? A. 3>2>1 B. 3>1>2 C. 2>3>1 D. 1>2>3…
A:
Q: What indicated hydrogen atom is the most acidic? B HAA H₂ of C CH3 D
A: We have to select the most acidic H
Q: CH3 NaOH, H20 I2 b) Rank the following compounds in order of increasing acidity: (b) CH3CH2OH (c)…
A: As you not specifiy so i am giving answer of 2 question A)this is a iodoform reaction in which…
Q: 2. Rank the following compounds from the least acidic to most acidic, provide a brief explanation,…
A: The decreasing acidity order is: C > A > B > D Compound C is most acidic. Compound…
Q: Rank the compounds in each group in order of increasing basicity.
A:
Q: 13) Rank these acids from least to most acidic SH HN A) DBCA (M.A.) B) DBAC (M.A.) C) BDAC (M.A.) D)…
A: Hello. Since more than one question has been asked, the first question shall only be attempted in…
Q: Rank the compounds in each group in order of increasing acidity.
A: The ease of losing H+ and thereby forming a stable anion would make the acid amongst the choices to…
Q: он SO3H COOH SO3H ČH3 ČH3 NO2 ČH3 III IV I II
A:
Q: Q8:- Arrange the compounds of each set in order of basicity:? Explain your choice ? Aniline…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Rank the following compounds in order of increasing basicity. The first compound in your ranking…
A: The given compounds are the conjugate bases of the acid. Now, the more stable conjugate base would…
Q: 2. Which carboxylic compound of each pair would you expect to be stronger acid? Indicate (a) or (b)…
A: The compound in which negative charge will be more stable will be more acidic in Nature. Hence…
Q: True or False C6H5CH2NH2 is the strongest base among the :following C6H5NH3 (a) p-NH2C6HANH (b)…
A: The correct statement is given below
Q: which compound is the stingey acid CH3-E - NH2 a. CH-č-o-H b. C. CH,-0-H
A: Since your question has multiple sub-parts, we will answer the first three sub-parts for you. If you…
Q: Which compound of each of the following pairs is the STRONGER BASE? (A) (B) O Option A O Option B…
A:
Q: Rank the following compounds in order of increasing acidity of their mostacidic hydrogen atom. (See…
A: The compound C is the least acidic because the electronegativity of nitrogen is less than that of…
Q: Rank the compounds in each group in order of increasing basicity. NH2 NH2 NH2 а. NHз -NH2 -NH2 O,N…
A: According to Lewis theory the compound which can donate lone pair of electrons is called base, and…
Q: Rank the following compounds in order of decreasing acidity. CH4 F3C-S HO. H3C-S- HO- HO, A C A: […
A: Solution : The acid-base reaction involves the donation of a proton from an acid to a base. This…
Q: Since each molecule of acetic acid which ionizes produces 1 H+ ic concentration in the solution are:…
A: Weak acid ionized partially. Degree of ionization (a) = 1.34 % = 0.0134 initial concentration of…
Q: Rank the following compounds in order of increasing basicity: NH₂ NH₂ NH₂ NH₂ A) B) D) OCH3 NO₂ 2
A:
Q: What is the correct rank of the following compounds in order of decreasing acidity? NH2 HO II IV…
A: Acidity of any molecule is directly proportional to the stability of -ve charge formed after…
Q: Arrange each group of compounds in order of increasing acidity.butyric acid, a@bromobutyric acid,…
A: Factors affecting on acidity of molecules- Electronegativity- Acidity increase with…
Q: HOO hydroxide acetate t-butoxide ethoxide The most basic of the ions is The least basic of the ions…
A:
Q: Which factors impact the relative stability of the two indicated bases, and in what way? O2N A
A:
Q: Rank the following compounds from strongest acid to weakest acid
A: pKa value for the given compounds are,
Q: Which is a stronger acid? a. HCl or HBr b. CH3CH2CH2NH3 or CH3CH2CH2OH2 c. CH3CH2CH2OH or…
A: To find: Which is stronger acid?
Q: Rank the labeled protons (Ha–Hc) in mandelic acid, a naturally occurring carboxylic acid in plums…
A: The stability of a conjugate base formed after the removal of a proton decides the acidic strength…
Q: Rank the following compounds in order of increasing acidity.
A: Given compounds:
Q: Rank these hydrogens as to acidity. H;C HO. нн A C A most, C least O C most, B least O A most, B…
A:
Q: -NH3 NH3 но- -NH3 H3C- -NH3 I II III IV
A: Acidity completely depends on the stability of conjugate base of that acid. If it is most stable…
Q: Rank the following compounds in order of increasing basicity: CH3O- , H2N- , H2O and NH3
A:
Q: (ii) Arrange the following compounds A - C in order of increasing basicity, and explain the reasons…
A:
Q: Rank the following compounds in order of increasing acidity. CF3 `OH H3C OH H3C´ ОН II II HO HO-…
A: Substituents that exerts -I effect increases acidity.
Q: Rank the following species in order of increasing basicity. Explain your reasons for ordering them…
A: According to Bronsted-Lowry acid base theory the compounds which have ability to donate proton or…
Q: 4. Rank the following compounds in terms of increasing basicity: Br
A: The less electronegative the element, the less stable the lone pair will be and therefore the higher…
Q: Solve the Attachment
A: Acids tend to lose H+ and form conjugate base. Stronger the acid, more is the Ka value and smaller…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Sort the compounds below in order of increasing acidity. Explain your answer.Regarding to the following compounds: NH3, H2O, H2SA) Rank the following compounds in order of increasing acidityB) Mention their conjugate bases and rank according to increasing their basicity order2-Hydroxybutanedioic acid occurs naturally in apples and other fruits. Rank the labeled protons in 2-hydroxybutanedioic acid in order of increasing acidity.
- Rank the following species in order of increasing acidity. Explain your reasons for ordering them as you do. HF NH3 H2SO4 CH3OH CH3COOH H3O+ H2OLabel each molecule according to increasing acidity with 1 as the weakest and 4 as the strongest based on their structure. Explain.Rank the bases below in order of increasing basicity.
- Propoxide (CH3CH2CH2O- ) is a larger molecule than ethoxide (CH3CH2O- ), yet they are equally basic. Explain why they are equally basic.Rank the following bases in order of increasing basicity: LDA, cyclohexane magnesium chloride, NaOme, benzylamine, water.Rank the compounds in each group in order of increasing acidity.

