Rank the following atoms in order of increasing first ionization energies (i.e., lowest to highest): Rb, F, Mg, B, N. A) F < N < B < Mg < Rb B) Rb < Mg < F B > N > F > Mg
Rank the following atoms in order of increasing first ionization energies (i.e., lowest to highest): Rb, F, Mg, B, N. A) F < N < B < Mg < Rb B) Rb < Mg < F B > N > F > Mg
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter6: The Periodic Table And Atomic Structure
Section: Chapter Questions
Problem 6.67PAE
Related questions
Question
100%
so confused on this question
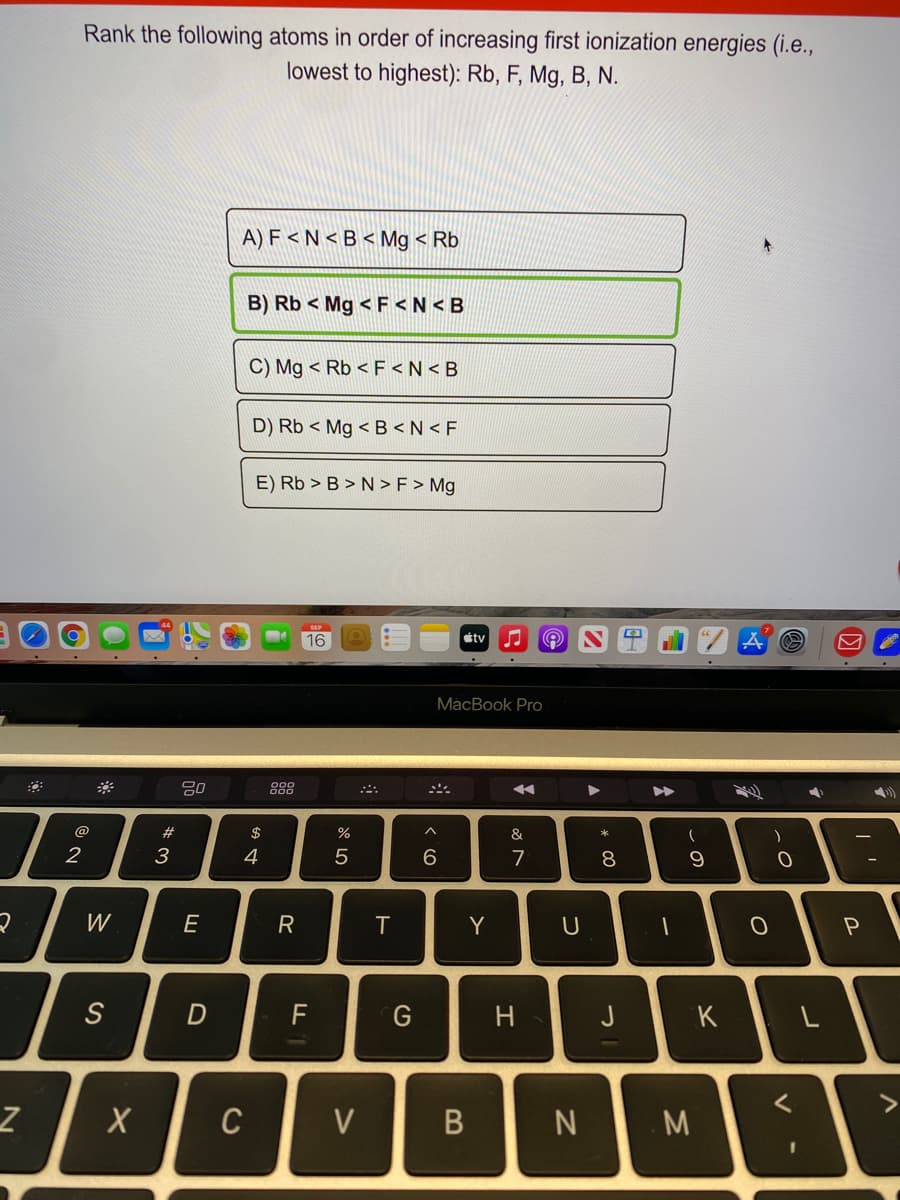
Transcribed Image Text:Rank the following atoms in order of increasing first ionization energies (i.e.,
lowest to highest): Rb, F, Mg, B, N.
A) F < N < B < Mg < Rb
B) Rb < Mg < F <N < B
C) Mg < Rb < F <N < B
D) Rb < Mg < B <N < F
E) Rb > B > N >F > Mg
16
tv
МaсBook Pro
80
888
@
#
$
%
&
2
3
4
6.
7
8
W
E
R
T
Y
S
G
H
J
K
C
V
M
B
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning