Rank the following compounds in order of increasing acidity, putting the least acidic first. CH3COOH FCH2COOH CICH2COOH BRCH2COOH I II III IV A) I
Rank the following compounds in order of increasing acidity, putting the least acidic first. CH3COOH FCH2COOH CICH2COOH BRCH2COOH I II III IV A) I
Chapter20: Carboxylic Acids And Nitriles
Section20.SE: Something Extra
Problem 20VC: Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the...
Related questions
Question
100%
please answer the 2 questions

Transcribed Image Text:23. Rank the following compounds in order of increasing acidity, putting the least acidic
first.
CH3COOH
FCH2COOH
CICH2COOH
BrCH2COOH
I
II
III
IV
А) I<IV< I<Ш
В) I<I<IV <I
С) I<Ш IV<I
D) II< IV < III< I
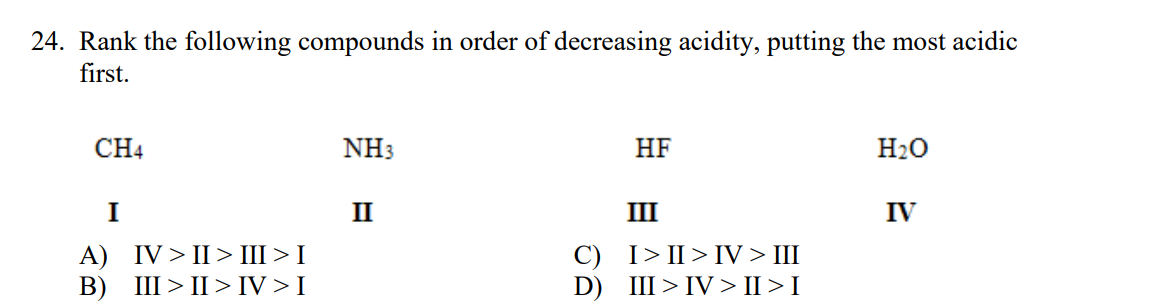
Transcribed Image Text:24. Rank the following compounds in order of decreasing acidity, putting the most acidic
first.
CH4
NH3
HF
H20
I
II
III
IV
IV > II> Ш>I
C) I>II>IV > III
D) III>IV > II > I
A)
В)
III > II > IV >I
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning