Rank the following from least amount to greatest amount present as product under the shown reaction conditions: КОМе I. II. III. IV. O IV. < III. < |I. < I. O I. < II. < IV. < III. O IV. < I. < II < III. O I. < III. < IV. < II.
Rank the following from least amount to greatest amount present as product under the shown reaction conditions: КОМе I. II. III. IV. O IV. < III. < |I. < I. O I. < II. < IV. < III. O IV. < I. < II < III. O I. < III. < IV. < II.
Chapter12: Gravimetric Methods Of Analysis
Section: Chapter Questions
Problem 12.31QAP
Related questions
Question
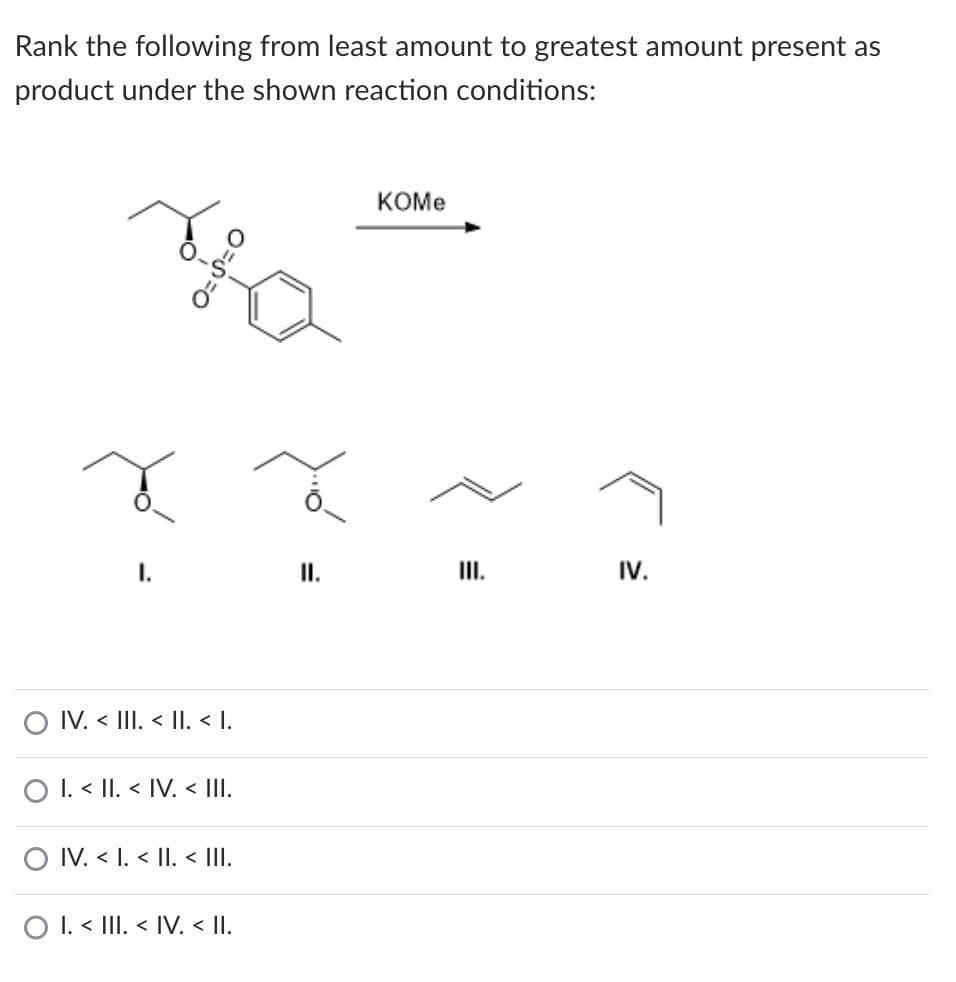
Transcribed Image Text:Rank the following from least amount to greatest amount present as
product under the shown reaction conditions:
КОМе
I.
III.
IV.
O IV. < III. < |I. < I.
O I. < II. < IV. < II.
O IV. < I. < II. < III.
O I. < III. < IV. < II.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole


Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole