Rank the samples of gas described in the table below in order of increasing average speed of the atoms or molecules in them. That is,select "1" next to the sample in which the atoms or molecules have the slowest average speed. Select "2" next to the sample in which th molecules have the next slowest average speed, and so on. gas sample average speed of atoms or molecules 1.9 mol of argon gas at 1.9 atm and -18. °C (Choose one) 1.4 mol of helium gas at 2.3 atm and 4. oC (Choose one) 2.6 mol of krypton gas at 2.8 atm and - 18. °C (Choose one) 1.1 mol of argon gas at 2.5 atm and 4. °C (Choose one) X Submit Assigr Continue Terms of Use 2019 McGraw-Hill Education. All Rights Reserved MacBook Pro F1 F10 DII F9 F8 F7 F6 O00 F5 F4 F3
Rank the samples of gas described in the table below in order of increasing average speed of the atoms or molecules in them. That is,select "1" next to the sample in which the atoms or molecules have the slowest average speed. Select "2" next to the sample in which th molecules have the next slowest average speed, and so on. gas sample average speed of atoms or molecules 1.9 mol of argon gas at 1.9 atm and -18. °C (Choose one) 1.4 mol of helium gas at 2.3 atm and 4. oC (Choose one) 2.6 mol of krypton gas at 2.8 atm and - 18. °C (Choose one) 1.1 mol of argon gas at 2.5 atm and 4. °C (Choose one) X Submit Assigr Continue Terms of Use 2019 McGraw-Hill Education. All Rights Reserved MacBook Pro F1 F10 DII F9 F8 F7 F6 O00 F5 F4 F3
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter8: Properties Of Gases
Section: Chapter Questions
Problem 104QRT
Related questions
Question
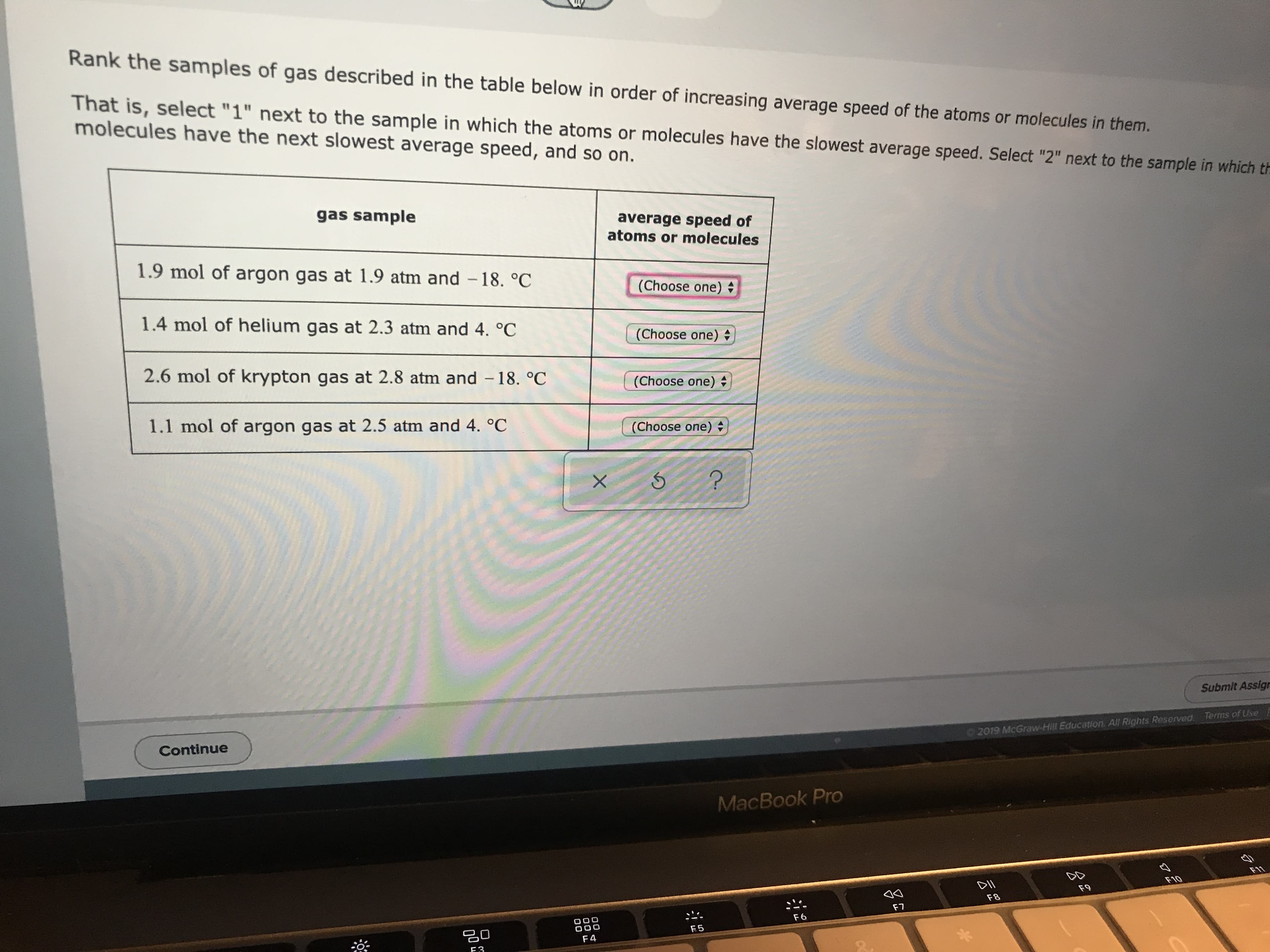
Transcribed Image Text:Rank the samples of gas described in the table below in order of increasing average speed of the atoms or molecules in them.
That is,select "1" next to the sample in which the atoms or molecules have the slowest average speed. Select "2" next to the sample in which th
molecules have the next slowest average speed, and so on.
gas sample
average speed of
atoms or molecules
1.9 mol of argon gas at 1.9 atm and -18. °C
(Choose one)
1.4 mol of helium gas at 2.3 atm and 4. oC
(Choose one)
2.6 mol of krypton gas at 2.8 atm and - 18. °C
(Choose one)
1.1 mol of argon gas at 2.5 atm and 4. °C
(Choose one)
X
Submit Assigr
Continue
Terms of Use
2019 McGraw-Hill Education. All Rights Reserved
MacBook Pro
F1
F10
DII
F9
F8
F7
F6
O00
F5
F4
F3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning