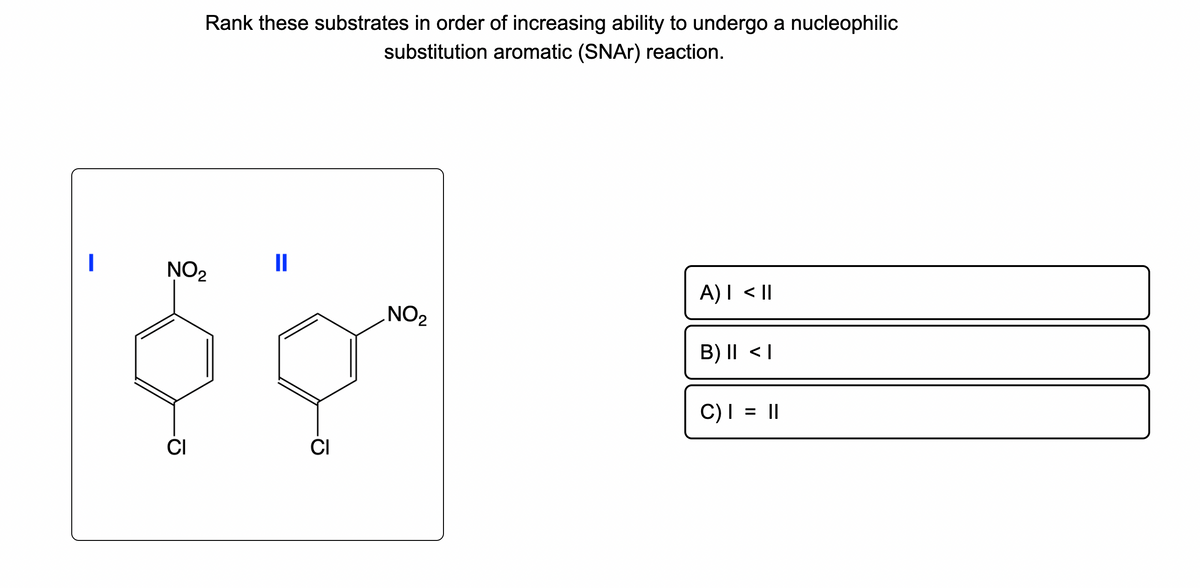
Rank these substrates in order of increasing ability to undergo a nucleophilic substitution aromatic (SNAr) reaction. NO₂ II $9 CI CI NO₂ A) I < II B) II
Q: 1 2 Which electrostatic potential map best represents the NaCl molecule? Question 13 ( 2 1 2 3 2 3…
A: The electrostatic potential map indicates the electron density on the surface of the molecule. Red…
Q: D Question 1 From the reaction above, which of the following is INCORRECT about Compound A (diene,…
A: Given are diene and dienophile.Diene and dienophile react to form cyclic product, This reaction…
Q: Drag the tiles into the numerator or denominator to form the expression. Each reaction participant…
A: chemically equilibrium in a reversible reaction is the state at which both forward and backward…
Q: Preparation of NaOH Solution: Calculate the volume of 6 M NaOH needed to prepare 500 mL of 0.1 M…
A: Use the dilution law to calculate the volume of 6M NaOH required to prepare a 0.1M NaOH solution.
Q: Specify the types of strain that destabilize the conformer shown below. Hint: All bonds not…
A: Given is structure of organic compound.Name of given compund is,2-methylbutane
Q: The step that completes the citric acid cycle is the conversion of malate (M) to oxaloacetate (OA)…
A: Answer:In any type of cell, oxidation always takes place at anode and reduction always takes place…
Q: Rank the following compounds in decreasing order of acidity. O Br ABCDE B. C. D. E. OA OB OK OD .E…
A: Given question related to the arrangement of carboxylic acid derivatives in decreasing order of…
Q: If the reaction 2H₂S(g) → 2H₂(g) + S₂(g) is carried out at 1065°C, Kp = 0.0120. Starting from pure…
A:
Q: Consider the reaction: C(s) + O₂(g) CO₂(g) Write the equilibrium constant for this reaction in terms…
A: For a reversible reaction, after some point in time the rate of the forward reaction becomes equal…
Q: Explain why the reaction is SN1 and not SN2
A: Given compound is, 1-bromo-1-phenylethane.Reagent given is NaCN
Q: Consider a nanogram and a microgram. Fill in the following sentence to best describe their…
A:
Q: Starting with the unbalanced equation for the combustion of ethanol, C₂H5OH(1)+O2(g)-CO₂(g)+?H₂O(1)…
A: Unbalanced reaction equation isC2H5OH (l) + O2 (g)→ CO2(g) + H2O (l)In a balanced chemical…
Q: An electron in an atom is known to be in a state with magnetic quantum number m₁ = 1. What is the…
A: n = principal quantum number Azimuthul quantum number, l = 0 to (n-1) Magnetic quantum number, ml =…
Q: Decide whether these proposed Lewis structures are reasonable. proposed Lewis structure : 0: H-O-H…
A: Lewis structure represents the arrangement of valence electrons of an atom in a compound. Lewis…
Q: Carbon-hydrogen bonds exhibit a range of different chemical reactivity that depends on molecular…
A: The carbon attached with single carbon is called primary carbon.The carbon attached with two carbon…
Q: The effect of chlorofluorocarbons (such as CCl2F2(g)) on the depletion of the ozone layer is well…
A:
Q: Distinguish between strong and weak electrolytes in terms of the arrows in the reaction: which one…
A:
Q: Two compounds, A and B are formed from elements X and Y as follows: Compound A contains 1.00 g of X…
A: Answer:Law of multiple proportions says that when two elements combine to form more than one product…
Q: 5. Identify as R, S, or achiral. H₂C. CH3 HC C CH H3C-C-CH3 CH3
A:
Q: (21): The ionization energy for potassium is 4.34 kJ/mol. What would be the ionization energy for…
A: Ionization energy can be defined as the minimum amount of energy required to remove the outermost…
Q: Using curved arrows, show the mechanism for the acid-base reaction. Be sure to show all lone pair…
A: The acid is a species that gives the proton to another species. The base is a species that accepts…
Q: Part 1 1.00 mL of 0.466 MNat M Part 2 2.00 mL of 3.47 mMLICI mM Part 3 5.00 mL of 5.55 mMZn²+ mM
A: We have to calculate the molarity of the given solution. Molarity is the moles of solute dissolved…
Q: There are different ways to measure pH given in your textbook. From the options listed below,…
A: Answer:pH tells us about the power of hydrogen ion concentration in the solution and its value is…
Q: Two Compounds: 3-methylbutyric acid and 4,5-dimethyldecane Suppose you took your two compounds,…
A: The given experiment represents the separation of two compounds, 3-methyl butyric acid, and…
Q: Figure 6-1 Hel A Br → B H Ha B 4 ܠ ܐ ܐ CI CIB 4 N 4 Br H # R Cl 4 ܐ It BY
A:
Q: How much heat in kilojoules is absorbed when 84.88 grams of Carbon Tetrachloride (CC14) is melted at…
A: Information
Q: Write the IUPAC name for the molecule shown in the Newman projection. Note for advanced students:…
A: In order to specify the IUPAC name for the given molecule in Newman projection we have to convert…
Q: OMe OMe NO₂ NO₂ HCI 40 °C A A A
A: For the first reaction diene reacts with HCl at 40°C this leads to the 1,4-addition of conjugated…
Q: 1. Draw all resonance structures for each of the following molecules or ions. Be sure to include…
A: When all the properties of a molecule cannot be explained by a single structure, we draw more than…
Q: Which compound is a tertiary alcohol? O3-hexanol O2-methyl-2-hexanol O 1-propanol…
A: We will draw all the given alcohol and find out the tertiary alcohol from them.
Q: Draw structural condensed formulas for all the constitutional isomers of C5H12
A: Given,The formula of the molecule: C5
Q: . Use stepwise synthesis including reagents and intermediates to complete the ollowing multi-step…
A: An organic reaction is one that involves the reaction of different reagents with the organic…
Q: Calculate strain energy for the conformer pictured below, using strain energy increments from
A: When there is 60 degree angle between two adjacent group it is calledgauche confirmation.
Q: Which of the following compounds are achirai? Br CH₁ -H -H HO- -OH Br 1 II, III, and IV I only II…
A: For any substance to be optically active or chiral, it must rotate the plane polarized light either…
Q: 20) Provide the IUPAC name for 2 of the following four molecules. Please provide an X in the…
A: Naming substituted alkanes and cycloalkanes with secondary SUFFIXES: Organic compounds containing…
Q: A 20.0 g block of iron (molar heat capacity 25.1 J/mol °C) at 83.4 °C is placed into 200.0 g of…
A: Answer:This question is based on law of conservation of energy where energy lost by hotter object…
Q: Provide the correct IUPAC name for AI(H₂PO4)3.
A: Al(H2PO4)3 is an inorganic salt with white Crystaline form. Here , Al = AluminumH= hydrogenP=…
Q: In which of the following species does sulfur have the lowest oxidation number? A) S²- B) Sa C)…
A:
Q: Express your answer as a chemical formula. View Available Hint(s)
A: To solve this problem, we have to write the molecular formula of a given 3D-model of a compound.
Q: Check t the table. x under each structure in the table that is an enantiomer of the molecule shown…
A: The chiral molecule that is non-superimposable on its mirror image is known as enantiomer and…
Q: How many intermediates are in the energy profile diagram below? Energy reaction coordinate
A: Intermediates in reaction energy profile diagrams are lies between two transition states.
Q: A chemist adds 140.0 mL of a 0.514M aluminum sulfate (Al, (SO₂),) & solution to a reaction flask.…
A: Millimoles of aluminium sulfate =?
Q: Cryolite, Na, AlF6(s), an ore used in the production of aluminum, can be synthesized using aluminum…
A: The reactant species which is fully consumed during the reaction is known as the limiting reagent.…
Q: The combustion of propane may be described by the chemical equation C₂H₂ (g) +50₂(g) → 3 CO₂(g) +…
A: C3H8(g) + 5O2(g) ----> 3CO2(g) + 4H2O(g)mass of C3H8(g) reacts = 88.0 gmass of O2(g) needed =…
Q: Which of the following are possible products of the reaction between hydrogen bromide and 2-pentene?…
A:
Q: Does larger dispersion forces increase or decrease boiling and melting point
A: Larger dispersion forces, also known as London dispersion forces or van der Waals forces, typically…
Q: Only typed explanation otherwise leave it
A: Catalytic hydrogenation is the addition of hydrogen molecule to the unsaturated compound in the…
Q: Preparation of NaOH Solution: Calculate the volume of 6 M NaOH needed to prepare 500 mL of 0.1 M…
A: Initial molarity of NaOH solution (M1) = 6 M Final molarity of NaOH solution (M2) = 0.1 M Final…
Q: Question 22 What is the systematic (IUPAC) name for the following alkane? % to
A: -Select the parent chain with the longest possible continuous carbon chain with a maximum number of…
Q: At 1000 degrees celsius Copper has an equilbirum vacancy conentration of 1 in 10^5 atoms. The…
A: The equation for the equilibrium concentration of vacancies can be used to calculate the enthalpy of…
Rank these substrates in order of increasing ability to undergo a nucleophilic substitution
Step by step
Solved in 3 steps with 3 images

- For each reaction, predict what mechanism will account for the major product(s) formed and write (SN1, SN2, or NR) in the boxes provided. Explanations or drawings are not required.can someone help me with this? Make a comparison table between the reaction mechanism SN1 and SN2, take into account thereaction order, nature of the substrates, solvents used, nature of the nucleophile, stereochemistry,kinetics, and other characteristics that you consider relevant.Complete the following reactions with the majority products and the missing reaction conditions. Indicate stereochemistry if applicable. Also indicate the type of reaction that took place (SN1, SN2, E1, E2, E1bc)
- please explain clearly and in detail. thank you! 2-butene reacts with NBS yields and forms two products, 1-bromobut-2-ene and 3-bromobut-1-ene. explain how this happens using mechanisms.The synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)HELP ASAP! please write out mechanism and reagants/reactants for this
- Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.About: Substitution vs. Elimination Reactions Note: Identify the major and minor product (s) that are expected for each of the following reactions.Complete the reactions given below, write down the type of mechanism (SN1, SN2, E1, E2)?

