Reagent Step in ProcedureObservations Data collected Mass: 3.0437 Pinacol Measured on balance Sulfuric acid added Heat Distillation (start) Distillation (finish) Volume: 5.0 750 EPM 150°C 750 RPM 250°C Pinacolone Purification Mass:\.4644 IR NMR 2,4-DNP Test Solid yellow color
Reagent Step in ProcedureObservations Data collected Mass: 3.0437 Pinacol Measured on balance Sulfuric acid added Heat Distillation (start) Distillation (finish) Volume: 5.0 750 EPM 150°C 750 RPM 250°C Pinacolone Purification Mass:\.4644 IR NMR 2,4-DNP Test Solid yellow color
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter33: Automated Methods Of Analysis
Section: Chapter Questions
Problem 33.4QAP
Related questions
Question
100%
Can you answer all parts to this question
- Theoretical yield of pinacolone in grams and moles
- show your calculations
- Final yield of pinacolone (grams and % yield)
- show your calculations
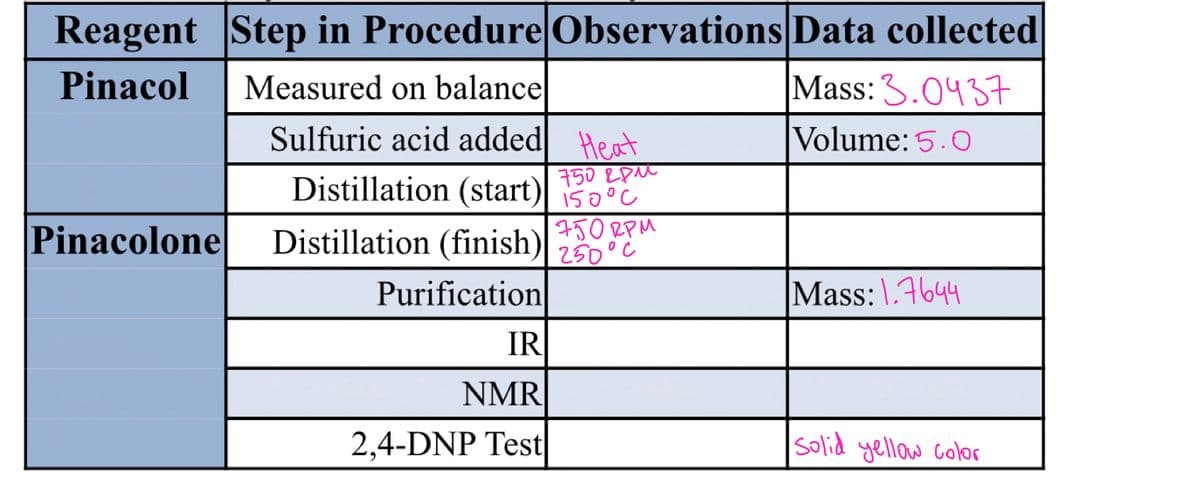
Transcribed Image Text:Reagent Step in Procedure Observations Data collected
Mass: 3.0437
Pinacol
Measured on balance
Sulfuric acid added| Heat
Volume: 5.0
750 EpM
Distillation (start)| iš0°c
150°C
Pinacolone
Distillation (finish)| 250°c
750 RPM
Purification
Mass:\.4644
IR
NMR
2,4-DNP Test
Solid yellow color

Transcribed Image Text:Part 1: Formation of pinacolone
Add 3.0 g of pinacol and 30 mL of deionized water to a 100 mL round-bottom flask.
Add a stir bar and clamp the flask to a ring stand, with the flask hovering 2-3 in above the
hot plate. Turn on the stirring function on the plate until the mixture is evenly moving.
Then slowly, carefully, add 5.0 mL of concentrated sulfuric acid. This is an exothermic
reaction. Allow the mixture to stir for 10 minutes.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
