Q: I want the mechanism for this “Nef reaction”
A: Nef Reaction: Reaction involving the conversion of nitro compound into carbonyl compound.
Q: Find the pH of 0.25M KF. Given Ka (HF) = 6.9x10-4 Show all work, use an ICE table and equation.
A:
Q: A 15.0 mL solution of Ba(OH)₂ is neutralized with 27.2 mL of 0.200 M HCl. What is the concentration ...
A: Welcome to bartleby !
Q: Draw both condensed and line structures corresponding to the following IUPAC names:(a) 3-Methylhept-...
A: Bond line structure is the one in which covalent are represented with line for each level of bond or...
Q: Consider the reaction of solid aluminum iodide and potassium metal to form solid potassium iodide an...
A: Reaction: Solid aluminum iodide and potassium metal to form solid potassium iodide and aluminum meta...
Q: Polymorphism can exist in any crystalline material, not just molecular solids. True or false? Explai...
A: Crystalline solids are those where a long-range order is present in the arrangement of constituent p...
Q: Consider the reaction of solid aluminum iodide and potassium metal to form solid potassium iodide an...
A:
Q: According to the following reaction, how many grams of barium hydroxide are required for the complet...
A: Stoichiometry gives the relationship between the amount of reactants and products in a balanced chem...
Q: Salsalate, which is an ester formed by the reaction of two molecules of salicylic acid, is another s...
A: Esters are formed by the reaction of acids or acid derivatives such as acid chloride and acid anhydr...
Q: Draw the structure of A, an intermediate in the synthesis of theantipsychotic drug risperidone. Expl...
A: Risperidone is an antipysichotic drug, it is used to treat bipolar, mental disorder. This medication...
Q: Explain why Xe, and to a limited extent Kr, form com-pounds, where as He, Ne, and Ar do not.
A: He, Ne, Ar, Kr and Xe all are of group 18 elements, they are also known as noble gas elements due to...
Q: Define Characteristic Spectroscopic Absorptions of Aldehydes and Ketones ?
A: The characteristic Spectroscopic Absorptions of Aldehydes and Ketones have to be given.
Q: Nitroglycerine ( C, H,N,0,) is a very strong explosive that degrades over time and is too shock sens...
A: Number of moles is calculated by using the formula: Number of moles=Given massMolar mass .....(...
Q: Consider the following reaction: A + B --> products. The rate law was found to be rate = k[A][B]^...
A: The order of Reaction refers to the relationship between the rate of a Chemical Reaction and the con...
Q: What kinds of reactions are common to alkanes? List an example of each?
A: alkanes are saturated hydrocarbon with no functional groups such as -COOH, double bonds present in i...
Q: (13) A 9.50 % by mass solution of acetone (C3H&O) in water has a density of 0.9849 g/mL at 20C. What...
A:
Q: Give A Summary of the Reagents for Reduction ?
A: There are many reducing agents which reduces different type of compounds. I will be discussing some ...
Q: 4. Identify each of the following salts (ionic compounds) when dissolved in water as generating an a...
A: 4 Different types of salts given: 1. Lithium cyanide (LiCN) 2. Potassium sulfate (K2SO4) 3. Aluminum...
Q: Which is NOT true about a reaction under reflux? O For reactions in which the reaction rate must be ...
A: Reflux: Reflux is a process where substance is heated up to its boiling temperature but the vapour i...
Q: Chemistry question
A:
Q: Heat a pinch of sodium chloride and sucrose in a separate dry and clean test tube for twominutes. Le...
A: After heating a compound there is some physical and chemical changes .
Q: At 0°C a 1.00L flask contains 5.00x 10-2mol of N2and 1.50x 10-2mol of O2. Calculate the partial pres...
A:
Q: Give the structure of a dimethylcyclobutane that is chiral
A: When all the four group attached to a carbon is different then it is termed as chiral carbon Chiral ...
Q: Calculate the heat evolved by 4.5 g of propane from the equation C, He + 502 3CO2 4 H,0 From the fol...
A: Heat of reaction or enthalpy change ∆Hrxn is defined as heat released and absorbed during a reaction...
Q: A student is to prepare a 250.-mL solution of 1.0 M glucose (C6H12O6, 180.16 g/mol). a) How many gra...
A: Given: The volume of the solution is 250 mL. Molarity of glucose solution =1.0 M Molarity can be c...
Q: The question below is on acid-base equilibria. Suggest an acid and a base whose titration curve wou...
A: Curve A, represents the pH titration curve of strong acid with weak base. Because, on addition of w...
Q: Why are intermolecular forces generally much weaker than bonding forces?
A: The reason for the relative weakness of intermolecular forces compared to bonding forces is also rel...
Q: A.) Determine the reaction order for this reaction. B.) Determine the rate constant for this reactio...
A: When the concentration of A is kept constant and the concentration of B is increased from 0.04 to 0....
Q: Change each of the following common names to a correct IUPAC name. (a) Isopropylamine(b) diethylamin...
A: IUPAC name the systematic name given to organic compounds which are considered same everywhere. Thes...
Q: a. What pH would you make the water layer to cause the carboxylic acid to dissolve in the water laye...
A: a The pKa value of acetic acid is 5 The carboxylic acid must be in charged form to be solvable in wa...
Q: If 26.0 g of NaOH is added to 0.800 L of 1.00 M Zn(NO₃)₂, what mass in grams of Zn(OH)₂ will be form...
A: Number of moles = mass/molar mass Molarity = moles/volume(in L) Moles = molarity × volume(in L) M...
Q: Using condensed structural formulas, write a balanced chemical equation for the reaction: addition o...
A: Condensed structural formula The formula in which the structure is drawn in lines are called Condens...
Q: A (green), B (blue), and C (red) are structural isomers as shown as figure .The molecular filmstrip ...
A: The explanation and the answer is as given below:
Q: Chemistry question
A:
Q: Rank the following substances in order of their expected SN1 reactivity. A) CH3CH2Br B)CH2=CHCH3Br C...
A:
Q: Write detailed answers to the following.a. Discuss the structure of diborane.b. Explain how silicon ...
A:
Q: Using the experimental data at 1/2 the equivalence point volume, calculate the Ka of acetic acid. Vo...
A: When acetic acid reacts with NaOH, the following reaction occurs: CH3COOH+NaOH→CH3COONa+H2O
Q: Consider a 0.10 M solution of a weak polyprotic acid (H2A) with the possible values of Ka1 and Ka2 g...
A: Given, Concentration of H2A, [H2A] = 0.10M a. Ka1 = 1.0 X 10-4; Ka2 = 5.0 X 10-5b. Ka2 = 1.0 X 10-4;...
Q: Given the reaction has a percent yield of 88.8%, what is the mass in grams of aluminum iodide that w...
A: 3I2 +2Al→2AlI3 % yield = 88.8% 2 moles of Al gives 2 moles of AlI3 26.98g of Al gives 407.6g of AlI3...
Q: How many ices are thene in D.I87 mol of Na kons
A: Since you have asked multiple question, we will solve the first questionfor you. If you want any spe...
Q: How many moles of H₂O can be formed from 2.87 × 10²³ molecules of NH₃ from the following equation? 4...
A: In the balanced Chemical Reaction - 4 NH₃(g) + 5 O₂(g) → 4 NO(g) + 6 H₂O(g) 4 moles of Ammonia give...
Q: A 35.0 mL solution of NaOH is neutralized with 23.4 mL of 0.250 M HCl. What is the concentration of ...
A: Solution C1V1= C2V2 where, C1, C2 - concentrations V1, V2 - volumes Given, C1= 0.250 M V1= 35 ml, V...
Q: Consider the collection of nonmetallic elements O, P, Te,I, and B. (a) Which two would form the most...
A: Hello, I am answering the first three sub-parts only. Please repost the question and mention which s...
Q: Identify C, the product of an SN2 reaction in the synthesis of raloxifene, adrug used to reduce the ...
A: The SN2 is the bimolecular nucleophilic substitution reaction. The rate-determining step of the reac...
Q: How are the elements in the periodic table arranged based on their increasing atomic number? AVOID P...
A: The term atomic number can be described as the no. of protons which a nucleus is having in an atom. ...
Q: Does entropy increase or decrease in the following process? N2 + 3H2 → 2 NH3
A:
Q: The negative terminal of a voltage source is connected to an electrode of an electrolytic cell. Is t...
A: The negative terminal of a voltage source is connected to an electrode of an electrolytic cell. Is t...
Q: How do phase transitions take place on the microscopic scale?
A: To find: How do phase transitions take place on the microscopic scale
Q: You need to make an aqueous solution of 0.147 M copper(II) chloride for an experiment in lab, using ...
A: Molarity = number of moles/volume of solution (in L) Moles = molarity × volume(in L) Number of mol...
Q: Protons and neutrons are found in the (a, surrounded by nearly empty space where (b) are located. (c...
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want...
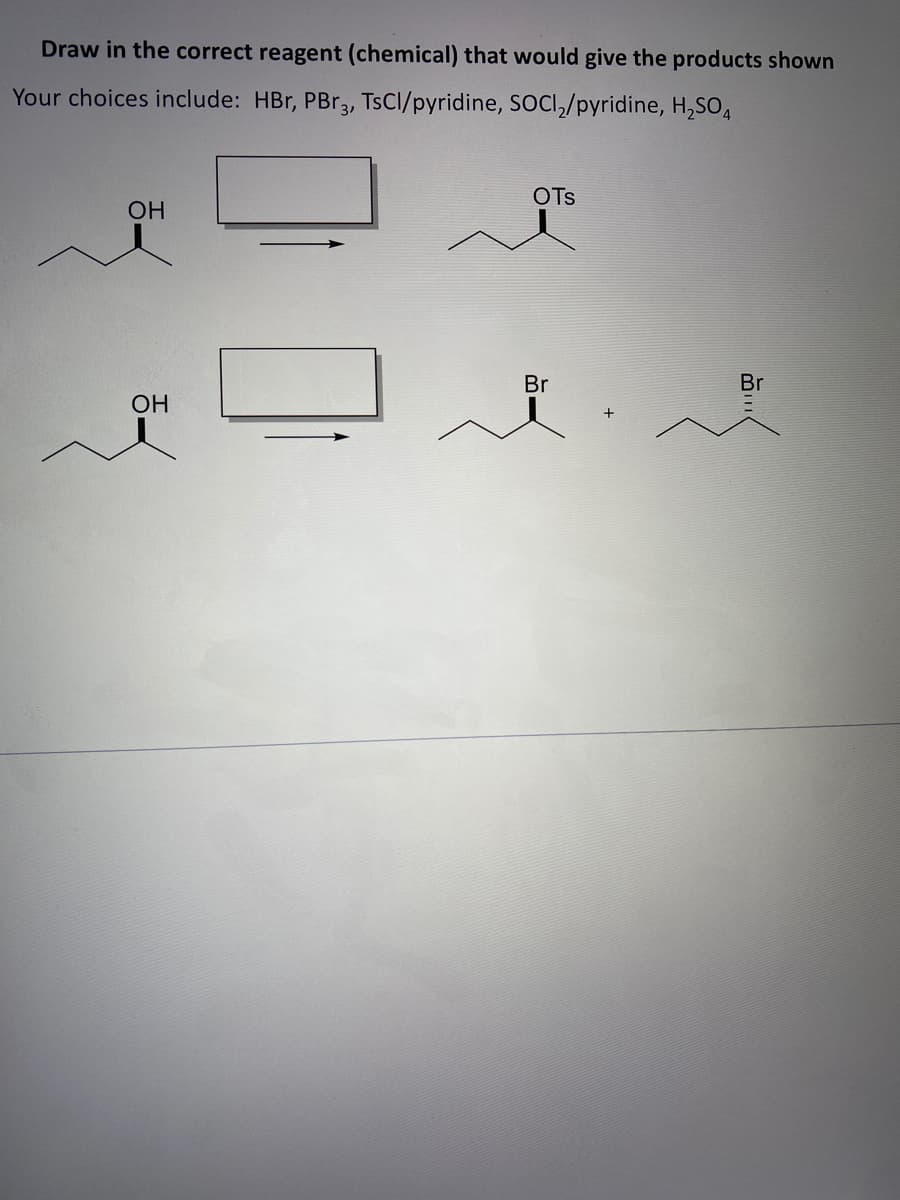
Step by step
Solved in 2 steps with 1 images

- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) is treated with each reagent. a. HCl b. CH3COCl c. (CH3CO)2O d. excess CH3I e. (CH3)2C = O f. CH3COCl, AlCl3 g. CH3CO2H h. NaNO2, HCl i. Part (b), then CH3COCl, AlCl j. CH3CHO, NaBH3CNKMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanoneDraw the major product of the reaction between 1-butanol and Na2Cr2O7, H2SO4, H2O.
- Draw and name compounds that meet the following descriptions: (a) Three acid chlorides having the formula C6H9ClO (b) Three amides having the formula C7H11NODraw a resonance structure of the acetonitrile anion, -: CH2CN, and account for the acidity of nitriles.1. You have an alkyl halide and the product has a thiol group without an alkyl, what is the reagant? 2. What is the reaction of disodium sulfide and Na2S, what is its product? 3. You have a thiol compound that reacts with sodium hydroxide, what do you get? 4. You have an oxyrane reacting with sulfuirc acid and methanol, what do you get? 5. Have an alkene reacting with Br, what do you get?
- name and draw the product/s formed from the reaction of 3-methylpent-2-ene: 1. with HBr 2. with Cl2 3. H2O in the presence of H2SO4 4. ethanol in the presence of H2SO4 5. Br2, H2O 6. [1] 9-BBN; [2] H2O2, OH-Draw compound B by reacting 2,4-dimethylcyclopentane-1-carboxylic acid with SOCl2Indicate the product of each reaction below
- The Ka1 of ascorbic acid is 7.94 x 10-5. Would you expect ascorbic acid dissolved in blood plasma (pH 7.35–7.45) to exist primarily as ascorbic acid or as ascorbate anion? Explain.(A true story.) A drug user responded to an ad placed by a DEA informant in a drug-culture magazine. He later flew fromColorado to Maryland, where he bought some 1-phenyl-2-propanone (P2P) from the informant. The police waited nearlya month for the suspect to synthesize something, then obtained a search warrant, and searched the residence. They foundthe unopened bottle of P2P; apparently, the suspect was not a good chemist and was unable to follow the instructions theinformant gave him. They also found pipes and bongs with residues of marijuana and cocaine, plus a bottle of methylamine hydrochloride, some muriatic acid (dilute HCl), zinc strips, flasks, and other equipment.(a) Assume you are consulting for the police. Show what synthesis the suspect was prepared to carry out, to provideprobable cause for the charge of attempting to manufacture a controlled substance.(b) Assume you are a member of the jury. Would you convict the defendant of attempting to manufacture a…From the given two compounds in the image, which reagents would differentiate the pair? a. Br2 in CH2Cl2 b. CrO3, H2SO4, acetone c. concentrated HCl with ZnCl2 d. aqueous FeCl3 e. aqueous NAHCO3 f. ammoniacal AgNo3



