Recall that for a lone pair to be involved in resonance, it must be in a p orbital, which requires the atom it is on to be sp² or sp hybridized. This means that for a situation with resonance, the actual hybridization of the atom is determined from the resonance structure where that atom has the lowest steric number (so it can use the required number of p orbitals to form pi bonds as shown in some of its resonance structures). This could be the situation in your initial drawing, or in one of the resonance structures you draw from it. All of the oxygens in the following molecules are sp² hybridized except one. Which of the following structures has an sp³ hybridized oxygen atom? НО. (A) (B) (D)
Recall that for a lone pair to be involved in resonance, it must be in a p orbital, which requires the atom it is on to be sp² or sp hybridized. This means that for a situation with resonance, the actual hybridization of the atom is determined from the resonance structure where that atom has the lowest steric number (so it can use the required number of p orbitals to form pi bonds as shown in some of its resonance structures). This could be the situation in your initial drawing, or in one of the resonance structures you draw from it. All of the oxygens in the following molecules are sp² hybridized except one. Which of the following structures has an sp³ hybridized oxygen atom? НО. (A) (B) (D)
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter7: Molecular Structures
Section: Chapter Questions
Problem 102QRT: There are two compounds with the molecular formula HN3. One is called hydrogen azide; the other is...
Related questions
Question
100%
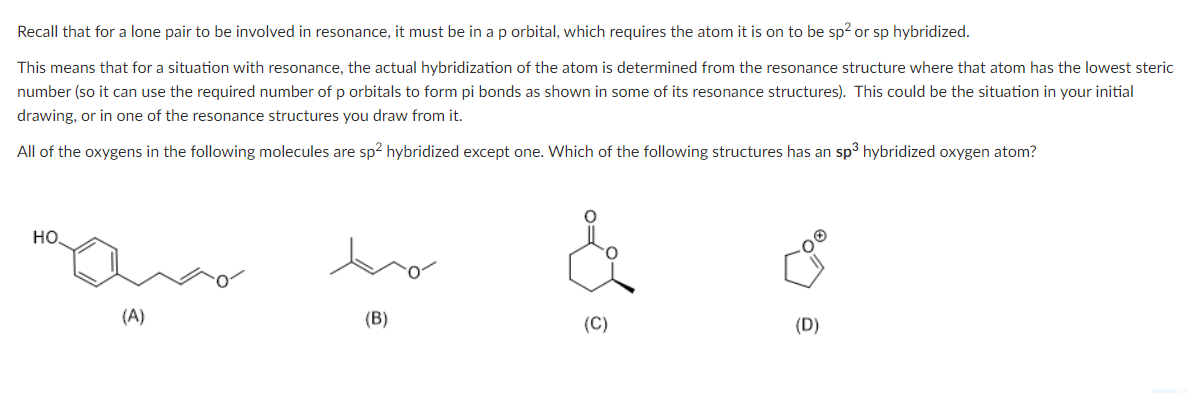
Transcribed Image Text:Recall that for a lone pair to be involved in resonance, it must be in a p orbital, which requires the atom it is on to be sp² or sp hybridized.
This means that for a situation with resonance, the actual hybridization of the atom is determined from the resonance structure where that atom has the lowest steric
number (so it can use the required number of p orbitals to form pi bonds as shown in some of its resonance structures). This could be the situation in your initial
drawing, or in one of the resonance structures you draw from it.
All of the oxygens in the following molecules are sp² hybridized except one. Which of the following structures has an sp³ hybridized oxygen atom?
НО.
(A)
(B)
(D)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning