Refer to the attached photo for the following question If a blue litmus paper is dropped to a solution containing these two reagents, what is the expected result? (Assume that the amounts are stoichiometrically proportional). A. The blue litmus paper turns purple. B. The blue litmus paper turns red. C. The blue litmus paper decolorizes. D. The blue litmus paper stays the same.
Refer to the attached photo for the following question If a blue litmus paper is dropped to a solution containing these two reagents, what is the expected result? (Assume that the amounts are stoichiometrically proportional). A. The blue litmus paper turns purple. B. The blue litmus paper turns red. C. The blue litmus paper decolorizes. D. The blue litmus paper stays the same.
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter8: Reactions In Aqueous Solutions
Section: Chapter Questions
Problem 3STP
Related questions
Question
Refer to the attached photo for the following question
If a blue litmus paper is dropped to a solution containing these two reagents, what is the expected result?
(Assume that the amounts are stoichiometrically proportional).
A. The blue litmus paper turns purple.
B. The blue litmus paper turns red.
C. The blue litmus paper decolorizes.
D. The blue litmus paper stays the same.
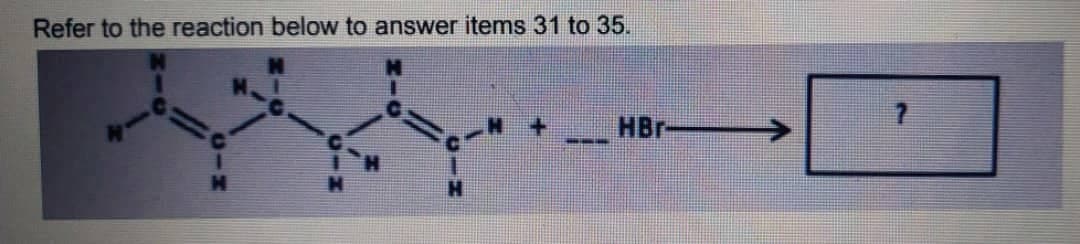
Transcribed Image Text:Refer to the reaction below to answer items 31 to 35.
HBr
---
CIH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

