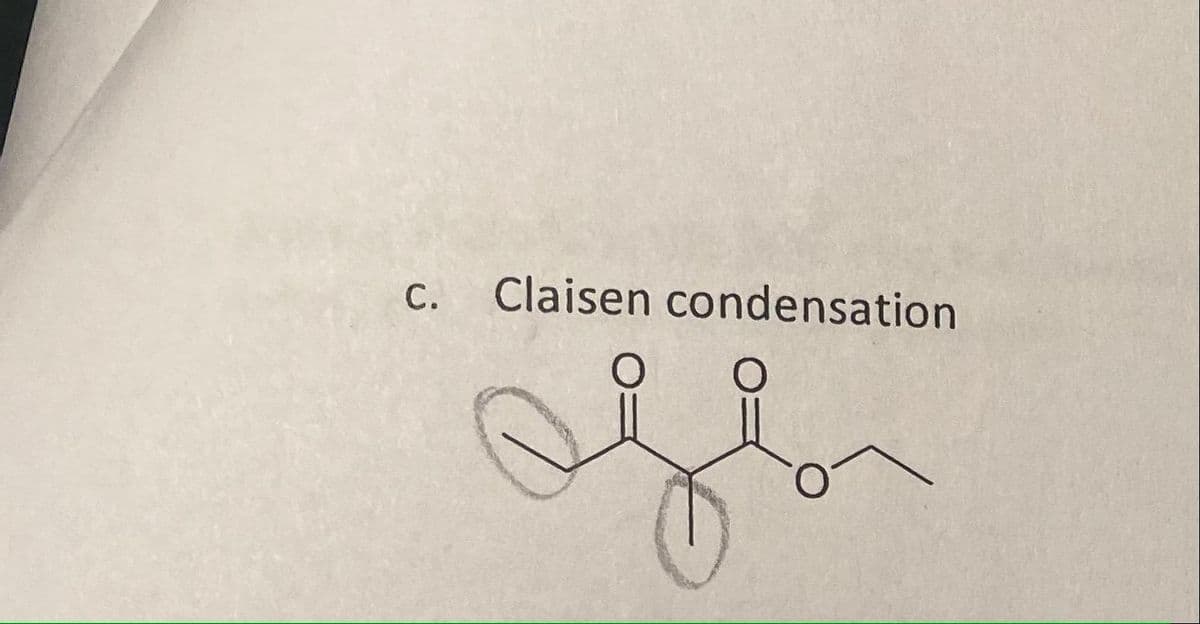
Q: Synthesize the following compound via Claisen condensation and any other necessary reagent.
A:
Q: Wellbutrin (1) is an antidepressant used to treat major depressive disorder. Add the missing…
A: The answer to the following question is-
Q: Explain the Reaction of ROH with PBr3—An SN2 Mechanism
A: A Ncleophilic Substitution reaction in which the rate determining step involves 2 components. 1. SN2…
Q: OH Br
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Draw the major product of this reaction. Ignore inorganic byproducts, methanal, or C2.
A: 1. OsO4 (osmium tetraoxide) is used to syn-dihydroxylation of alkenes 2. NaIO4 is oxidising agent,…
Q: MAKE: CEN CEN FROM: or USING ANY REAGENT WITH 42 CARBONS (SHOW STEPS)
A: GIVEN:-
Q: Devise a synthesis of attached compound from the indicated starting material.
A: The synthesis of given compound from the indicated starting material can be shown as follows,
Q: Show two different methods to synthesize alcohol A using a Grignar reaction.
A:
Q: Show how this cyclic ether compound could be prepared from an alkyl halide compound in an SN2…
A:
Q: HO Br
A: Conversion of alkyl halide to grignard reagent Attack of grignard reagent as nucleophile to the…
Q: Devise a synthesis of attached compound from the indicated starting material.
A: The starting material of the synthesis of compound is phenol. The structure of the final product…
Q: Please help! Provide the reagent needed to complete each transformation
A: NOTE: please note according to our policy , we can only answer first three subparts of a question.…
Q: Can you help propose a sy
A: 1. Cyclohexane is converted in to cyclo-1,3-hexadiene : cyclohexane upon treated with chlorine…
Q: CN
A:
Q: Devise a synthesis of each compound from the indicated starting material:
A: Given: To prepare: cis-1,2- dimethoxycyclohexane from cyclohexene
Q: A nucleophilic addition is not considered SN1 or SN2 because: A.) The reaction proceeds through an…
A: Given, Nucleophilic addition is not considered as SN1 or SN2. Why? SN1 stands for unimolecular…
Q: d. Но Br
A:
Q: HO, HO
A: Dimethylfumarate after reduction with LiAlH4 gives butan-1,4-diol which is converted to…
Q: Using the specified starting material and any other reagents, show how each of the following…
A: When acetylene is treated with sodamide then sodium acetylide is formed ,and then it is treated with…
Q: arrange the following alcohols in order of increasing rate of reaction with Lucas reagent
A: Lucas reagent is a solution of anhydrous zinc chloride solution in hydrochloric acid (ZnCl2 + HCl).…
Q: Arrange the following alcohols in order of increasing rate of reaction with the Lucas reagent.
A: Lucas reagent is a solution made up of with Conc HCl + Anhydrous ZnCl2 It is used to distingiush…
Q: Bombykol is the sex pheromone of the silk moth.
A: Suzuki Coupling : It is a cross-coupling reaction between organic halide and organocarbon compounds…
Q: Show the process of synthesizing an enolate compound from cyclohexanone using an LDA catalyst.
A:
Q: HO3S- -CH3 Br CH3
A: Detail mechanistic pathway is given below to carry out the conversion
Q: Devise a stepwise synthesis of attached compound from dicyclopentadiene using a Diels–Alder reaction…
A: Our target molecule is
Q: он Synthesize the compound in 2 different ways using Grignard reagents and reactions
A: In the organic synthesis, suitable reagent converts the reactant into products. Reagents attacks…
Q: Provide a synthetic route for the following transformation: Please provide steps and type of…
A: Given reaction:
Q: Show all the arrow mechanism including the PROPER reagents.
A:
Q: Propose a synthetic route for the following conversion:
A: 1. Alochols when they treated with acid it gives alkene by elimination of water 2. Alkenes are…
Q: Show how the benzyl methyl ether can be synthesized from benzene:
A: To show the preparation of benzyl methyl ether from benzene:
Q: Provide necessary carbonyl compound and phosphonium ylide to yield the alkene. What type of ylide…
A: Given,
Q: ОН Br ОН
A: For synthesis, first we have to block acid and alcohol group as ester and ether respectively .…
Q: Br
A: Interpretation - To complete the following reaction and provide the reagent in the given reaction .…
Q: Please give me the reagents and show the mechanism. Thank you so much.
A: These reaction involes the Michael addition reaction. Michael addition reaction involves the…
Q: From the given starting material provide a reasonable synthetic route to the following plecule. O,N.
A: Here we have to prepare the given compound from the starting material 2-methylbuta-1,3-diene in…
Q: H
A: Applying concept of organic synthesis via wittig reaction and organometallic.
Q: Devise a synthesis of attached compound from the indicated startingmaterial. You may also use any…
A: The reactions involving organic compounds are called organic reactions. Electrophilic and…
Q: Please rovide a synthetic approach for the following reactions. Make sure to do the retrosynthetic…
A: Given that,
Q: use an organic reagents no longer than tw or three carbons or benzene to synthesis this compond
A:
Q: Devise a synthesis of attached alkene using a Wittig reaction to form the double bond. You may use…
A: 1). Oxidation of 2-propanol with PCCpyridine chlorochromate to give 2-propanone2).…
Q: он он H3C он HO, target compound starting material
A: Following is the mechanism of the product formation from the given starting material.
Q: How To Synthesize a Compound Using the Robinson Annulation ?
A: Given: Synthesize a Compound Using the Robinson Annulation
Q: OH
A:
Q: HO mplete the
A:
Q: Devise a synthesis of attached compound from the indicated starting material.
A: In this reaction, we have to convert one isomeric form to another isomeric form. For this, we have…
Q: Explain why it is not possible to prepare tert-butyl phenyl ether using a Williamson ether…
A:
Q: ? Br
A:
Q: From benzaldehyde, compunds of 2 carbons or less, and any inorganic reagent you want synthesize the…
A:
Q: Show how the p-cresol can be synthesized from benzene:
A: Benzene is a six member planar aromatic molecule which contains 6 pi electron. P-Cresol constitutes…
Q: What starting material is needed to synthesize each compound by a ringclosing metathesis reaction?
A: Ring closing metathesis : In order to obtain the ring closing metathesis (RCM) , Grubbs catalyst is…
Step by step
Solved in 2 steps with 2 images

- The generic structural formula for a 1-alkyl-3-methylimidazoliumcation is where R is a —CH2(CH2)nCH3 alkyl group. The meltingpoints of the salts that form between the 1-alkyl-3-methylimidazolium cation and the PF6- anion areas follows: R = CH2CH3 (m.p. = 60 °C), R = CH2CH2CH3(m.p. = 40 °C), R = CH2CH2CH2CH3 (m.p. = 10 °C), andR = CH2CH2CH2CH2CH2CH3 (m.p. = -61 °C). Why doesthe melting point decrease as the length of alkyl groupincreases?Explain why the configuration of a polymer of isobutylene is neither isotactic, syndiotactic, nor atactic.Polymer PXX has a glass transition temperature of 49°C. What is the typical appearance and mechanical behaviour of this polymer PXX?
- A student in New York City carried ou1t a boiling point determination for cyclohexane {b.p. 81• C) according to the procedure in this laboratory manual. Will this student's observed boiling point be the same as the value obtained by another student in Denver, Co'lorado.{Nicknamed the "Mile-High City" due to its high elevation)? Will it be lower or higher? Ex plain your conclusion.The vapor pressure of 1-chlorotetradecane at several temperatures are tabulated below. T (ºC) P∗ mm Hg 98.5 1 131.8 5 148.2 10 166.2 20 199.8 60 215.5 100 Use the calculated values for ln(P∗)ln(P∗) to estimate the vapor pressure at 185°C.1. What is the limitation of the sublimation laboratory technique? 2. When can sublination be applied as a process of separation?
- 11c. Nylon 6 and poly(ε-caprolactone) have similar polymer backbone structure with one carbonyl, 5 methylene and a heteroatom, nitrogen or oxygen, respectively. Each is able to undergo crystallization. However, their thermal transition temperature differs significantly. The glass transition temperature (Tg) for Nylon 6 is 52 °C, whereas the Tg for poly(εcaprolactone) is 112 degree lower than Nylon 6 at – 60 °C. Similarly, the melting temperature (Tm) for Nylon 6 is 223 °C, whereas the Tm for poly(εcaprolactone) is 60 °C. Briefly explain the significant differences in the thermal properties of these two polymers.Is a mobile phase composition 40:60 of acetone:hexane more polar than a mobile phase composition of 25:75 acetone:hexane?Briefly describe the technique for packing a capillary melting point-tube

