Q: 1. 2.
A:
Q: Specify whether the two structures are resonance contributors to the same resonance hybrid. Be sure…
A: The structures given are,
Q: D-o +) S.
A: We know that, According to Huckel's rule, an aromatic compound must have (4n + 2)π electrons that…
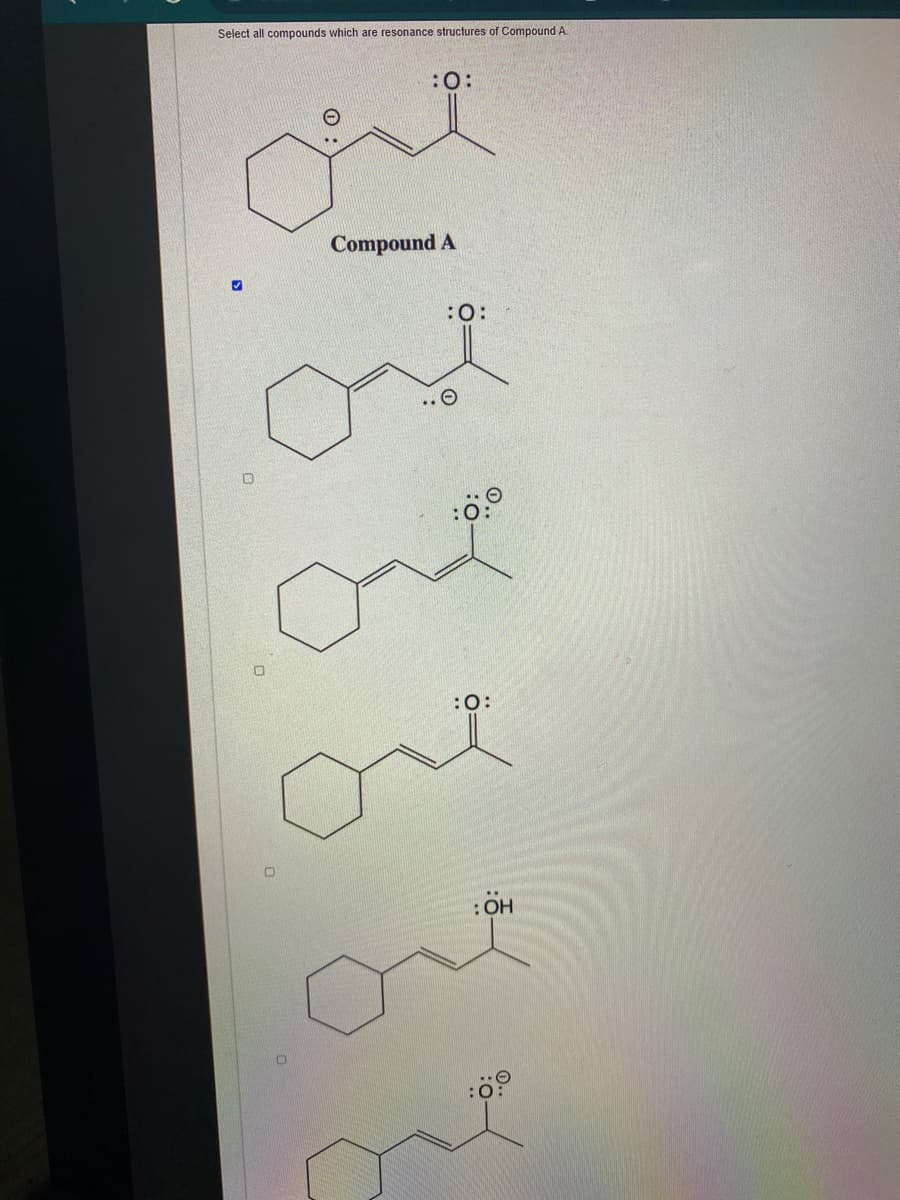
Q: Which of the following pairs depicts resonance structures? :0: :ö-H A) and H2N NH2 H2N NH CH3 CH3 B)…
A:
Q: The calicene molecule has a different dipolar moment of 0 and also the dipole points to the larger…
A:
Q: How many of the following elements to form compounds with an extended Octect Pb Kr Si B
A: Given elements, Lead (Pb) Krypton (Kr) Silicon (Si) Boron (B)
Q: НО :0:
A: Rp
Q: Identify
A: We will find out aromatic non aromatic and antiaromatic
Q: Please give the best resonance and second best resonance for SO2. Please explain why the best…
A: The stability of resinance structure is directly related with formal charges , octet completion and…
Q: uzet HO. No2 ONAY, coset Mith
A:
Q: How many additional resonance forms can be drawn for phenanthrene shown below?
A: Resonance structure is basically rearrangement of electron from one bond to adjacent bond but not…
Q: 6. Draw all significant resonance structures for the following compound, including curved arrows to…
A: Resonance structures of any compound are those structures which shows different possible Lewis…
Q: All possible resonance structure of THC (tetrahydrocannabinol)
A:
Q: a. Draw as many possible resonance structures of the molecule, (show the direction of electron…
A:
Q: :O: z: O:
A: To find: The resonance structure is given as,
Q: Draw as many resonance structures as you can for each of the following structure, and use…
A: Resonance structures arise when there exists a conjugated double bond/vacant orbital relative to an…
Q: identify which is the most and least basic among the given. refer to the number and stability of…
A: Here basicity means to donate the lone pair of electrons. The nitrogen atom contains lone pair of…
Q: 1. 2. H' H エ エ 3.
A: Note : As per our company guideline we can answer only three subparts. Kindly repost the question…
Q: 2. The two species shown are structurally very similar. Draw all significant resonance structures…
A:
Q: Between the following radicals; which one is more stable? I or II PHHC(*)CH2ET (1) PHCH2C(*)HEt (1I)…
A: Given free radicals are PhHC(•)CH2Et (I) PhCH2C(•)HEt (II) The free radical with more…
Q: Which of the these statements is correct? O All of these statements are correct. The greater the…
A: Correct option is A. All are correct. A.Greater the predicted stability of resonance contributor…
Q: Provide the bond line structure of a compound that is able to undergo Allylic lone pair and allylic…
A: Allylic lone pair: lone pair of an electron adjacent to a double bond. Allylic carbocation: An…
Q: Which structure is NOT aromatic? d. Do .. H3C :Br:
A: In order to decide a compound to be aromatic the following points are considered. The compound…
Q: H,N
A: We are having two organic molecule structures , we have to predict the common name as well as the…
Q: C HC- H2C- C +)
A: Resonance structures are a set of two or more lewis structures that collectively describe the…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided resonance structures,…
A:
Q: Classify the following compounds (if flat) as aromatic, antiaromatic, or nonaromatic.
A: For a compound to be aromatic, the compound must be flat, cyclic, completely conjugated, and follow…
Q: For each of the following groups of molecules (labeled A–E), rank the resonance forms in order from…
A:
Q: For the following compound identify the lone p dicate if each lone pair is localized or delocalize…
A:
Q: Give the formal charge on the highlighted carbon in each species. All H's and electrons on the…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3.
Q: Using the concept of aromaticity and resonance, determine which of the following compounds has the…
A: Due to the polarity of the carbonyl group, there is sepertation of charge in…
Q: Identify whether each structure, shown below, can participate in resonance and draw the associated…
A:
Q: Define the consequence of resonance stabilization ?
A: In case of certain molecules, the single Lewis structure can not explain the bonding and all…
Q: Resonance structure of ethyl propanate
A: Sometimes the chemical bonding of a molecule cannot be represented using a single Lewis structure.…
Q: How many resonance contributors are in the following molecule? H -C+ H.
A: The possible resonance contributors of benzyl carbocation are- I and V are different as bonds are…
Q: including arrows For the following structures, complete resonance structures electron flow and…
A: The ion given is,
Q: Based on the criteria stated above, classify the following compounds as aromatic, antiaromatic, or…
A:
Q: Explain with the help of resonance structures which of the two nitrogen atoms is more basic in the…
A:
Q: What is the other significant resonance form for the structure shown? Select one:
A: Resonance structures can be defined as the structures representing the delocalization of electrons…
Q: b) Draw 3 more major contributing resonance structures for this molecule. Of the 4, circle the most…
A: The resonance structure of a molecule describes the delocalization of electrons. However, all the…
Q: Which of the following resonance structures is the greatest contributor to the resonance hybrid? O:
A:
Q: Consider the following structure a) Use mechanistic arrows to construct the significant resonance…
A: By drawing conjugating strutures , we can determine the major and minor contributor.
Q: Draw all resonance contributors for each of the following molecules or ions. Be sure to include the…
A: NOTE- As the question contains multiple subparts, we will solve the first three subparts for you. To…
Q: The following molecule is
A: Huckel’s rule explains that if the compound obeys (4n +2) π electrons in conjugated system then it…
Q: =0 A В
A: The question is based on the concept of polarity. we have to determine which of the given molecule…
Q: Which of the following resonance structures has the greatest contribution to the hybrid? Explain. H.…
A: From the given resonance forms, which has greatest contribution to the hybrid has to be given.
Q: Cycloheptatrienone is an unusually stable ketone. Draw an aromatic resonance structure that explains…
A: we have to draw an aromatic resonance structure for cycloheptatrienone
Q: Draw resonance structures for the following compound: 요
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Draw all possible resonance structures for the following cation, and indicate which structure makes the largest contribution to the resonance hybrid.Which of the given resonance structures (A, B,or C) contributes most to the resonance hybrid? Which contributes least?Which of the given resonance structures (A, B, or C) contributes most to the resonance hybrid? Which contributes least?
- Which of the following resonance structures has the greatest contribution to the hybrid? Explain.Give a clear handwritten answer with explanation....give the all possible resonance structuresDraw all reasonable resonance forms for the following molecules. Hypothesize on which of the forms contributes most to the overall resonance hybrid in a couple of sentences for each compound.
- Why was the formal charge of (+) placed on this carbon? I understand everything else about the question but can someone help me with explaining this. Thanks!The anion drawn below displays resonance and has two main contributing structures. One ofthe two contributing structures is displayed. Draw the second contributing structure in theempty box below:All resonance structures with arrow. Major resonance?

