Q: 3.31 Which of the following ane meso compounds? Br Be Br CH, CH, C-C HO (a) (b) (e) Be CH, CH,
A: since you have posted multiple subpart question, we will answer the first 3 for you. Kindly post the…
Q: Draw the structure of TRIPHat pH 5. Show the stereochemistry of each Calpha chiral center.
A:
Q: calkulate the molkcular HO OH Weight 2 R O=C Where ( R) is : CH,COOH 3-6 CH2-N CH2COOH 3 H = 1,C= 12…
A: The compound given is,
Q: raw the structure of the major organic product of the reaction below. CH3CH2 CH2CH3 Zn(Cu) CH212…
A: When alkene react with Zn(Cu)/CH2I2.It is known as Simmons - Smith reaction .
Q: Is this a meso compounds?
A: Given compound,
Q: 3. a) Assign R or S designation to each of the following compounds. i OH ii H OH H,CH,C ii соон iv…
A:
Q: 5. 다음 거울상 이성질체 각 탄소의 R과 S를 결정하시오. (a) (b) (с) но. ОН H. „CH3 CH3CH2 H. H3C CH3 ----
A: We have to determine the R and S of each of the following given isomer in the mirror as follows in…
Q: 8-) Are the following compounds chira confiqura tion it is chiral and draw th each structure CH3 -9…
A: NOTE : Since you've posted multiple sub-parts,we'll solve first three sub-parts for you. To get the…
Q: Which of the structures shown below Is not aromatl2 Clck on a letter A to D to answer. CH B, D.
A: A compound is aromatic is it is Sp2 hybridised, cyclic and planar. and most importantly follows…
Q: 23. Draw the final product and identify R or S or racemic in all chiral centers in the product. Ph,…
A: Given that : We have to draw the final product and identify the R or S racemic in all the chiral…
Q: Which of the following molecules is (R,E)-1-fluoro-2-methylhept-2-en-4-ol? ÕH https://ibb.co/módYKJq…
A:
Q: a) 21. Which of the following is a pair of enantiomers? Circle appropriate letters. Alkanes Br Br b)…
A: Enantiomers The molecule which have non superimposable mirror images of each other are called as…
Q: what is the lbest mpound X o Give mechamism? to make Compound (9) oH (b) BY BY Br Nat
A: In this question we have to synthesis the compound X and also write its mechanism.
Q: 28) Label each asymmetric carbon in the molecule below as having the R or Sconfiguration. HO2C H.…
A: Given molecule:
Q: • Part C 2mathybutane Drag the appropriste labeis to their recpeotive targets. Labeic oan be uced…
A:
Q: For the given ee values, calculate the percentage of each enantiomer present. Q: 90% ee
A: Enantiomeric excess (ee) refers to the % or degree of excess enantiomer present in the given…
Q: खो जव पजो e ह Pि्ने कक्ट केकपलेपरर (मतु-तम (तम5ु)- ०H--CH,- CHO be the prodert it कैम्ट CH3-CH CCH3…
A: Detail mechanistic pathway is given below to find out the product for oxidation and reduction…
Q: c) Rank by number of stereoisomers. Use a "1" for the highest number, followed by a "2", then a "3"…
A:
Q: OH (R) (S) Он
A: A meso compound is an achiral compound with chiral centres. A meso compound possesses an internal…
Q: a) show the stereogenic centre(s) by labeling them with a star (*) b) determine the priorities of…
A: Chiral Compound have chiral centre and chiral centre is tetrahedral carbon which have all four…
Q: Is this a meso compounds?
A: Ki
Q: 1. Give the hybridization for the underlined carbon in PHCOOCH3: a. sp b. sp? c. sp d. sp 2.…
A: By the guidelines of bartleby i am answering first question only. Please repost the remaining…
Q: 19. Which compound is not a meso compound? H. (b) (a) CI CH CI CI CI H. (d) H CH (e) CI H Br CH Br
A: Applying concept of plane of symmetry to determination of meso compounds.
Q: 4) Assign R or S configuration to the following molecules: CH2CH2CH2 CH3 CH, Br CH2CH3 NH2 H3C…
A: Since you have posted a question with sub-parts , we are entitled to answer the first three…
Q: At -80 °C, Reaction of HBr with Butadiene, the transition state for the carbócation. A) stille B)1,4…
A:
Q: a. O3, CH;Cly, -78 °C b. Me,S b OH + enantiomer OH но H.
A: Given reaction is : Product of the reaction = ? Options are :
Q: compounds. Label pairs of enantiomers and label meso compounds. Slga anudomotaib ni oluelom onebvb d…
A:
Q: a) show the stereogenic centre(s) by labeling them with a star (*) b) determine the priorities of…
A: The question is based on the concept of stereochemistry. we have to assign stereogenic…
Q: Is this a meso compounds?
A: Meso Compound: Meso compounds are achiral compounds that has multiple chiral centers. The mirror…
Q: What arethe reagents needed to complete this reaction? a. Hz/ Lindlar's cataly st b. Li, E+NH2,-78°…
A: Welcome to bartleby ! We have to tell the appropriate reagent for the above transformation .
Q: Using the nomenclature guidelines given in class, give the correct IUPAC names for the following…
A: Compounds with OH attached to the cycloalkylring are named as cycloalkanol Compounds with CHO…
Q: Determine the absolute canfiguration each thiral center int pair of diastereonomen CH3 OH %2. OH Br…
A: The configuration on the chiral carbon is known as RS-nomemclature. Following rules are used to…
Q: Provide a name for each. Be sure to include stereochemistry in the name!! CH3 t -00- HO 114 Br H tat…
A: we have to the name the given structures
Q: Identify the indicated hydrogens in the molecules below as pro-R or pro-S. Ho Ha CO2H H3*N H…
A: • When a tetrahedral carbon can be converted into chiral center by changing only one of the…
Q: 3. Identify the stereocenters in the compounds below and assign each stereocenter as Ror S. Include…
A: For both compounds stereocentres and configuration has been assigned
Q: A 50:50 mixture containing the compounds below is expected to be optically inactive. !!!!!IC/ OH CI…
A: Optical activity refers to a property of a compound or mixture to rotate plane polarized light to…
Q: 2. Assign R or S configuration to the asymmetric carbons (designated by asterisk) in streptimidone…
A: The R/S is a system of absolute nomenclature of configuration. It follows a set of rules to identify…
Q: For the substituted cyclohexane compound shown, identify the atoms that will interact with the…
A: Concept - In cyclohexane, there are two positions - 1) axial 2) equatorial The groups present in…
Q: CI || OI OI and II O III ON "CI 8 & IV Which of the above has 2 stereogenic centers with both R…
A: we have to determine which of the given structures has R configuration for the 2 stereogenic centers…
Q: Identify the indicated faces in the molecules below as Re or Si. a `CH2CH3 b • side a: side b: a H.…
A: Re and Si faces: A face is labelled as Re if, when looking at that face, the…
Q: For the given ee values, calculate the percentage of each enantiomer present. Q: 99% ee
A: Given, ee = 99 %
Q: Make a model of R-1-chloro-1-methoxyethane CH3CH(OCH3)CI (let the light green atom be chlorine) Draw…
A:
Q: àchiral 3. LOCate the siereogenic ccnter () in each compound. OH OH OH O.) B) C:) OH CH OH
A: In this question, we will explain about the stereogenic center(s) in each compounds. How we will…
Q: what is the best mpound to make X o Give mechamism? compound OH (b) BY BY Br Nat
A:
Q: Which substituent has the highest priority? Br O a A O b.D Oc C O d. The molecule is not chiral. O…
A: Welcome to bartleby ! Introduction : We have to tell the priority of substitutent .
Q: CH CI H HOH CH3 CH HO H CLH CHs CH CH HOH 23 A pair of enantiomers of the following is: CHs TT TTT…
A: Given
Q: O || CH3CH₂-C-H A) CH3CH₂CH₂CN || C) CH₂CHCH CN HCN CN -? B) CH3CH₂CHC=NH OH D) CH3CH₂CHOH T CN
A:
Q: 18) Can the molecule shown below be properly described as a meso compound? a) b) c) CI CI CI CI
A:
Q: comprunds Y yhore th Compound X has paden uinonte , A= 271 (s) 8- 2-19 L) ond lompeund y has A= 2-77…
A: This problem is based on nmr spectroscopy
Q: 2 H ( A lily X IF Br
A:
From the given, identify the meso compound.
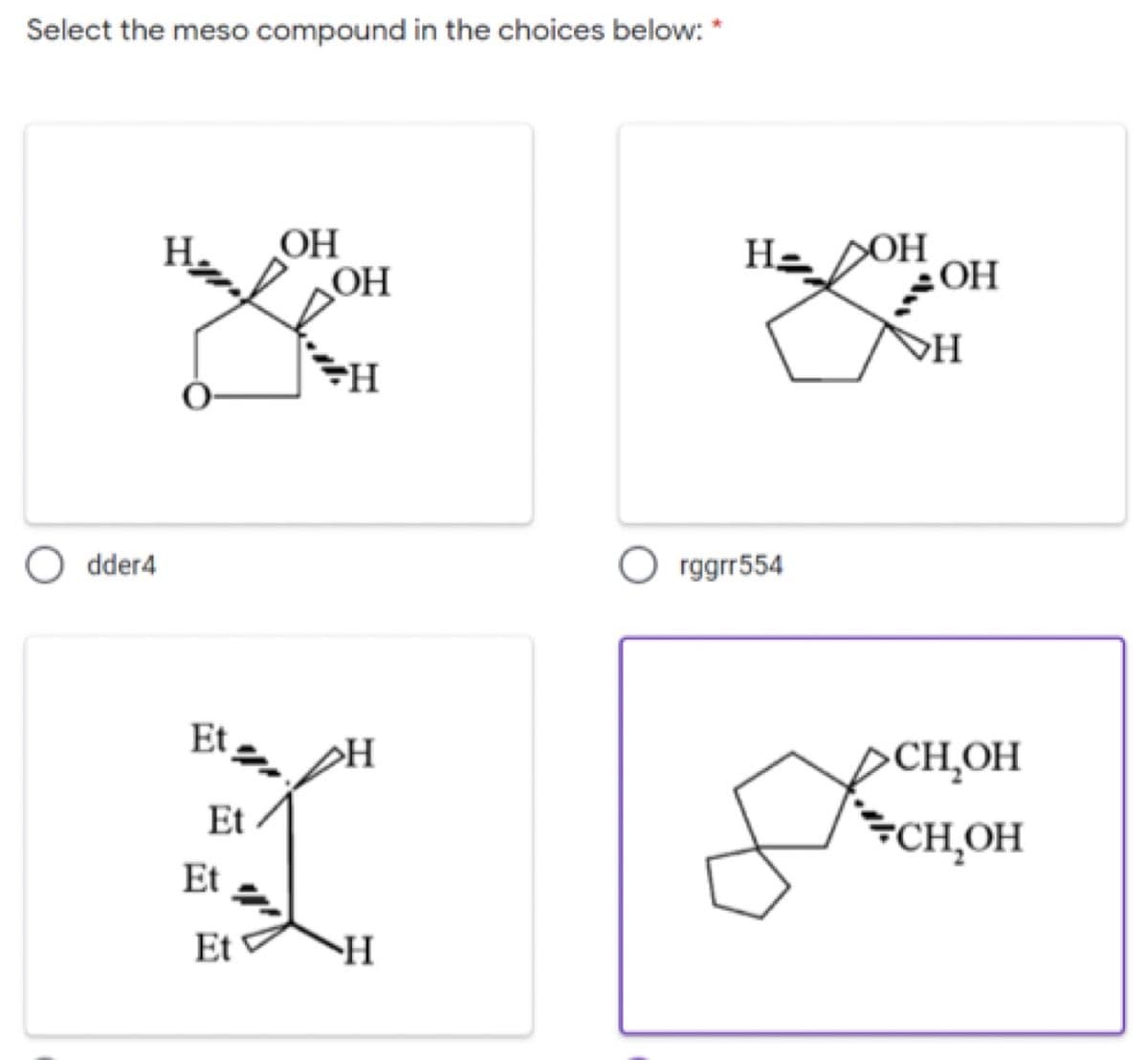
Step by step
Solved in 2 steps with 1 images

- Assign R or S configuartion to the following molecules.Which reaction below give a par of diastereomers and Why?Construct models of 1,2-dibromocyclopentaneand draw all the possible stereoisomers. Label each chirality centre with an*,andan Ror S. Indicate clearly which pairs are relatedasenantiomers and which as diastereoisomersor meso.
- Suppose we aempt the conversion of fumaric acid to deuterated malic acid with BD3·THF,followed by oxidation with D2O2in NaOD(aq). Show all the possible stereoisomers (as Fisherprojections) that may be formed, and draw the mechanistic pathways (showing stereochemistry)that lead to these possible productsIdentify the meso compounds from the followingDraw EI - MS for CH3CH2CH2CH 2BR Explaining your resonance for the peaks show clearly.
- Please explain. 5.38 asked which species were chiral, but I need help with 5.39. Thank you!Which configuration (R or S) does the bottom asymmetric carbon have for the d seriesof sugars? Which configuration for the l series?a. Idêntify thè cònfigûràtiòn of the dôublè bônd. b. Rèdraw the strùctùre and lábel the chîrál cèntérs usîng an ásterisk. c. Rèdràw the strùctüre with ALL thê chirál cèntèrs in S Cònfigûratîon. d. Dráw the ènantiômer of the strûcture dräwn in letter C. e. identify the number of diastereomers that this compound can have.
- pls give me answer of this question with explanation immiditely and i will rate you sir.Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the otherThe following Walden cycle has been carried out. Explain the results, and indicate where Walden inversion occurs.



