Select whether the property is likely to increase, decrease, or would have no effect in the given parameter.* Increase Decrease No effect Low Ka: Dissolution rate in aqueous solvent High pH : Excretion rate in alkaline urine Particle size reduction: Dissolution rate Low particle surface area: Dissolution rate Lipophilicity: Absorption across BBB High partition coefficient: Bioavailability Amorphous polymorph : Dissolution rate Drug-receptor ionic interactions :Affinity to receptors Conformational flexibility: Receptor selectivity Covalent bonding :Duration of action
Select whether the property is likely to increase, decrease, or would have no effect in the given parameter.* Increase Decrease No effect Low Ka: Dissolution rate in aqueous solvent High pH : Excretion rate in alkaline urine Particle size reduction: Dissolution rate Low particle surface area: Dissolution rate Lipophilicity: Absorption across BBB High partition coefficient: Bioavailability Amorphous polymorph : Dissolution rate Drug-receptor ionic interactions :Affinity to receptors Conformational flexibility: Receptor selectivity Covalent bonding :Duration of action
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.10QAP
Related questions
Question
100%
ANSWER THEM ALL FOR AN UPVOTE. I will downvote if not.
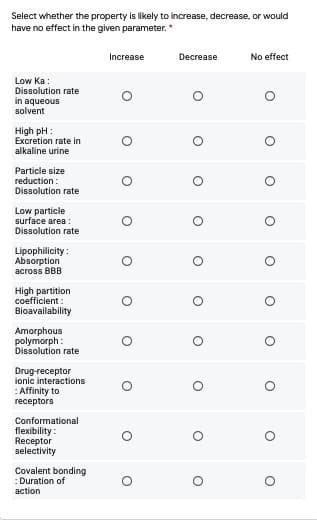
Transcribed Image Text:Select whether the property is likely to increase, decrease, or would
have no effect in the given parameter. *
Increase
Decrease
No effect
Low Ka:
Dissolution rate
in aqueous
solvent
High pH :
Excretion rate in
alkaline urine
Particle size
reduction:
Dissolution rate
Low particle
surface area :
Dissolution rate
Lipophilicity :
Absorption
across BBB
High partition
coefficient :
Bioavailability
Amorphous
polymorph:
Dissolution rate
Drug-receptor
ionic interactions
:Affinity to
rеceptors
Conformational
flexibility:
Receptor
selectivity
Covalent bonding
: Duration of
action
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning