Show reagents to convert bromocyclopentane to each of the following compounds. Br OH OH (c) ОН (f) C=CH (b) Br OH (a) (d) (i) OMe OH OH "OMe (k) HO H `H.
Show reagents to convert bromocyclopentane to each of the following compounds. Br OH OH (c) ОН (f) C=CH (b) Br OH (a) (d) (i) OMe OH OH "OMe (k) HO H `H.
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter19: Enolate Anions And Enamines
Section19.6: Acetoacetic Ester Synthesis
Problem 19.11P
Related questions
Question
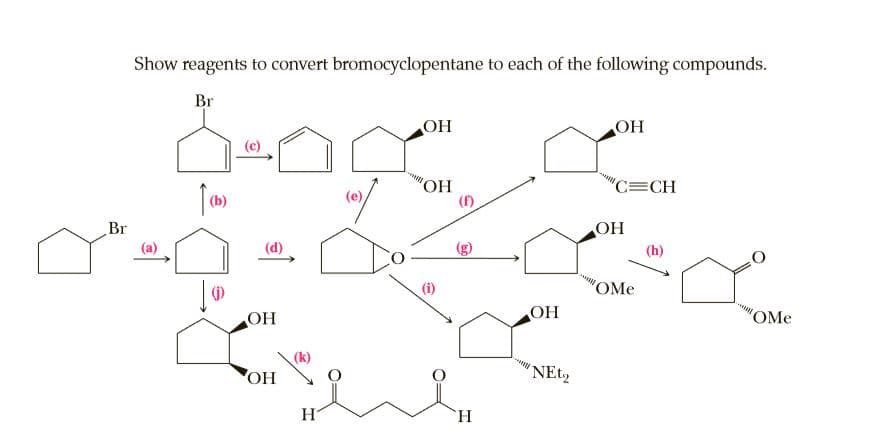
Transcribed Image Text:Show reagents to convert bromocyclopentane to each of the following compounds.
Br
OH
OH
(c)
ОН
(f)
C=CH
(b)
Br
OH
(a)
(d)
(i)
OMe
OH
OH
"OMe
(k)
HO
H
`H.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 4 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
