Q: Please transform and give starting material or product. Show stereochemistry and mechanism as…
A: In this question, we will give the product and Mechanism. You can see details Solution below.
Q: First, draw the structure of the alkyl bromide that will ONLY give the alkene shown in an…
A: Elimination reaction are those reaction where functional group is lost to form unsaturated compound…
Q: Choose the product of the following reaction.
A:
Q: Identify the cyclohexyl substrate which would undergo the fastest E2 with base.
A: E2 elimination is a one step concerted mechanism reaction in which dehydrohalogenation (or any other…
Q: Select the best reagents to effect the given transformation.
A: Answer : option B is the correct answer. When aldehyde react with NaBH4 in water it will leads to…
Q: What is the reagent R1 in this reaction sequence?
A: In the given reaction sequece first step aldehyde functional group is protected with ethylene glycol…
Q: Show the mechanism for this reaction using arrow pushing
A: Friedel craft alkylation Catalyst AlCl3 Carbocation stability…
Q: Show the complete mechanism for an E2 reaction with 1-bromo-2-tert-butylcyclohexane
A: In this question, we will draw Complete mechanism for E2 Reaction. You can see details Solution…
Q: Supply the structures of reactant(s) or product(s) for the following aldol and Claisen reactions
A:
Q: Provide the mechanism for the 1st reaction in this synthesis: КОН
A: We have to draw the mechanism of given reaction
Q: The by-product of the aldol reaction is not drawn here, what is it?
A:
Q: What is the target product formed after the following sequential reactions?
A:
Q: Draw curved arrows to show the movement of electrons in this step of the mechanism.
A:
Q: Please give the reagents that will perform the following reaction.
A:
Q: For the reaction shown, draw the transient product of one equivalent of reagent adding across the…
A: The given alkyne is converted into alkene (product A) in presence of 1 mol of H2,Pt
Q: Give a Grignard reation where the following reagent will participate
A: Alkyl magnesium halide (RMgX) is known as Grignard reagent. Grignard reagent is prepared by the…
Q: OEt OEt OEt
A: As this is a multipart question, only three parts are solved here. Please repost rest of these…
Q: scheme
A:
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone…
A: Given: product
Q: For the reaction shown, choose the most likely reaction pathway and draw the organic product.
A: This reaction can happen by Nucleophilic substitution reaction . Here azide ion as the nucleophile…
Q: Topose a synthesis of the aldehyde using ethane and propane as your only sources of carbon atoms.…
A: Since you have posted multiple questions in a session, as per guidelines we are entitled to answer…
Q: Construct a model of methycyclohexane with the methyl attached axially. Make the ring undergo…
A: We have given that Construct a model of methycyclohexane with the methyl attached axially. Make the…
Q: Show how you would accomplish the following synthetic conversions. More than one step be required.
A: In the above reaction, alkane is being converted to alcohol.
Q: React
A: Available Reagent is : NaH, THF CH3-Br This reaction is called " Williamson etherification…
Q: I need the reaction mechanism of this synthesis
A: Welcome to bartleby !
Q: Using
A: We will give mechanism of the reaction.
Q: Draw the products of the two step reaction sequence shown below. Ignore inorganic byproducts. If the…
A:
Q: Can you please draw me the synthetic route (not mechanisms) for this reaction by using…
A: Step I : conjugate addition of OH to unsaturated ketone. Step II : it is an haloform reaction in…
Q: down the product
A: Given:
Q: Show two different ways to make an epoxide from a trans alkene, one that gives the trans epoxide and…
A: Trans product :
Q: What is the retrosynthesis and synthetic strategy of the attached molecule? Please make the…
A:
Q: 3. Name other two reagents which are used for acetylation. Give mechanism for each reagent.
A: Acetic anhydride and acetyl chloride are two reagents for acetylation of aniline.Mechanism for…
Q: reagents
A: Friedel crafts alkylation reaction are mostly used in this question.
Q: Show the structure of the major and minor products of the following reactions:
A: Electrophilic aromatic substitution reaction where the C-H bond of aromatic ring are replaced by…
Q: Please help me draw the MAJOR product(s) of the this reaction.
A: It is an example of Friedal craft alkylation reaction
Q: Please provide a synthetic approach for the following reactions. Make sure to do the retrosynthetic…
A:
Q: in this E2 reaction what is the major product
A:
Q: Show the mechanism for the following transformation HO.
A:
Q: Provide a synthesis of the following target from the given starting material. from benzene
A:
Q: Question is attached
A:
Q: Please rovide a synthetic approach for the following reactions. Make sure to do the retrosynthetic…
A: Given that,
Q: Show all the arrow-push mechanism including the RIGHT reagents.
A:
Q: Show the retrosynthesis mechanism of this equation with curved arrows representing electron transfer…
A: Note : 1-Butene on hydroboration and oxidation produces the 1-butanol. Benzoic acid and 1-butanol…
Q: Draw the products of the three step reaction sequence shown below. Ignore inorganic byproducts. If…
A:
Q: Which of the following synthetic routes is the one that will most successfully generate a.
A: Alpha hydrogens are acidic in nature when base is used it is abstracted and gives conjugate base,…
Q: Question attached
A:
Q: provide a synthesis of the target compound shown from the starting material that is provided
A: The synthesis of the target compound shown from the starting material that is provided is given…
Q: what are the reagents, mechanism and reagents in this sequence, whilst talking about regioselective…
A: Favorskii rearrangement is base catalyzed reaction in which the alpha-halo carbonyl compound form…
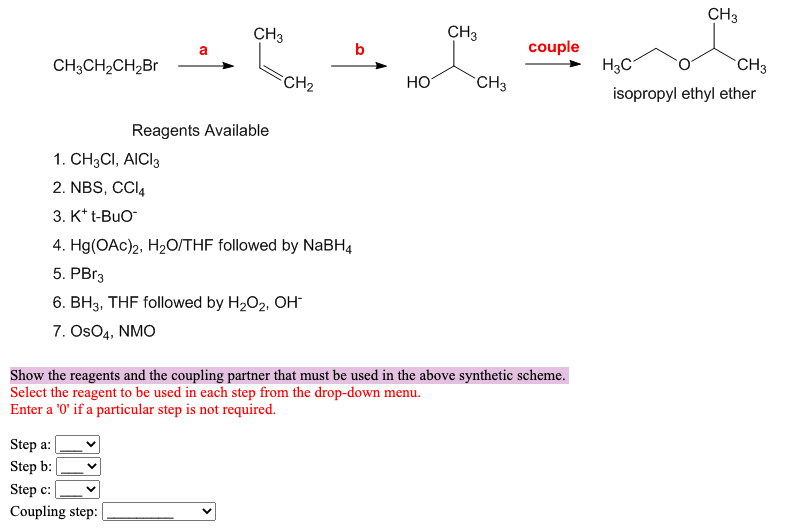
Show the reagents and the coupling partner that must be used in the above synthetic scheme.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- 1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the aboveOrganotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?(True/False) The use of sodium iodide in acetone promotes SN2 mechanism for alkyl halide resulting in the formation of insoluble salts in the mixture. (True/false) For alkyl halides, we use a solution of AgNO3 in ethanol because ethanol is a polar protic solvent suited for SN1 mechanism. (True/False) 1-bromobutane, 2-bromobutane (aka sec-butyl bromide), tert-butyl bromide, benzyl chloride, and bromobenzene will not form precipitates in either 1M NaI/acetone and 2% AgNO3/EtOH.
- tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate Typically yield 6.34g How would I calculate theoretical and percent yield for the reaction ? Please and thank you!tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g Why is the percent yield be lower than expected? 53.9% .. Is it due to evaporation? Is it not the major product? If this reaction was with 2-butanol/HCl instead of tert butanol, how would I obtain the products/mechanism, and determine major products? Thank you!!tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g How would I calculate theoretical and percent yield for the reaction ? Please and thank you!
- tert-butyl chloride (2-chloro-2-methylpropane) from tert- butyl alcohol (tert-butanol) using an acid catalyzed dehydration reaction Physical Constants Compound Mol. Wt (g/mol) Density (g/mL) b.p. (°C) m.p. (°C) 2-Methyl-2-propanol 74.12 0.786 82 25 2-Chloro-2- methylpropane 92.57 0.851 51 -25 Hydrochloric acid 37% soln in H2O 1.18 -85 -26 12.0ml of t-butyl alcohol, with 25.0ml 12.0M HCl, 12ml of water, 12ml of saturated aqueous sodium bicarbonate, 12ml of saturated aqueous sodium chloride, magnesium sulfate 1 (CH3)COH + 1 H3O^+ Cl^- -> 1 (CH3)3CCl + 2 H2O Typically yield 6.34g Why is the percent yield be lower than expected? 53.9% .. Is it due to evaporation? What is the major product of this reaction? isobutene or t-butylchloride? If this reaction was with 2-butanol/HCl instead of tert butanol, how would I obtain the products/mechanism (SN2?), and determine major products? If sn2, why? Thank you!! This was…Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Percent yield: 68 melting point:115-122 what is the lit melting point for this product? Did you successfully make pure 2,3-Diphenylquinoxaline? Explain
- Solution:- 3. Determine the amount, in of a (2.20 M s olution of dichloromethane needed to completely react with 15.72g cyclohexene to give 1,2-dibromocuclohexanw. Assume 12% excess is needed in order to react completely. a . How much 1,2 -dibromocyclohexane would theoretically be produced ? c. How many ML of the 2.20 M Br2 solution are required?Classify each solvent as protic or aprotic. a.(CH3)2CHOH b.CH3NO2 c.CH2Cl2 d.NH3 e. N(CH3)3 f.HCONH2Please complete reactions in clear handwritten of all subparts

