Q: Using the reagent list, identify the reagents needed for the three-step synthesis below. Note: there…
A: The Grignard reagent is an organometallic reagent. It is made up of magnesium metal and alkyl…
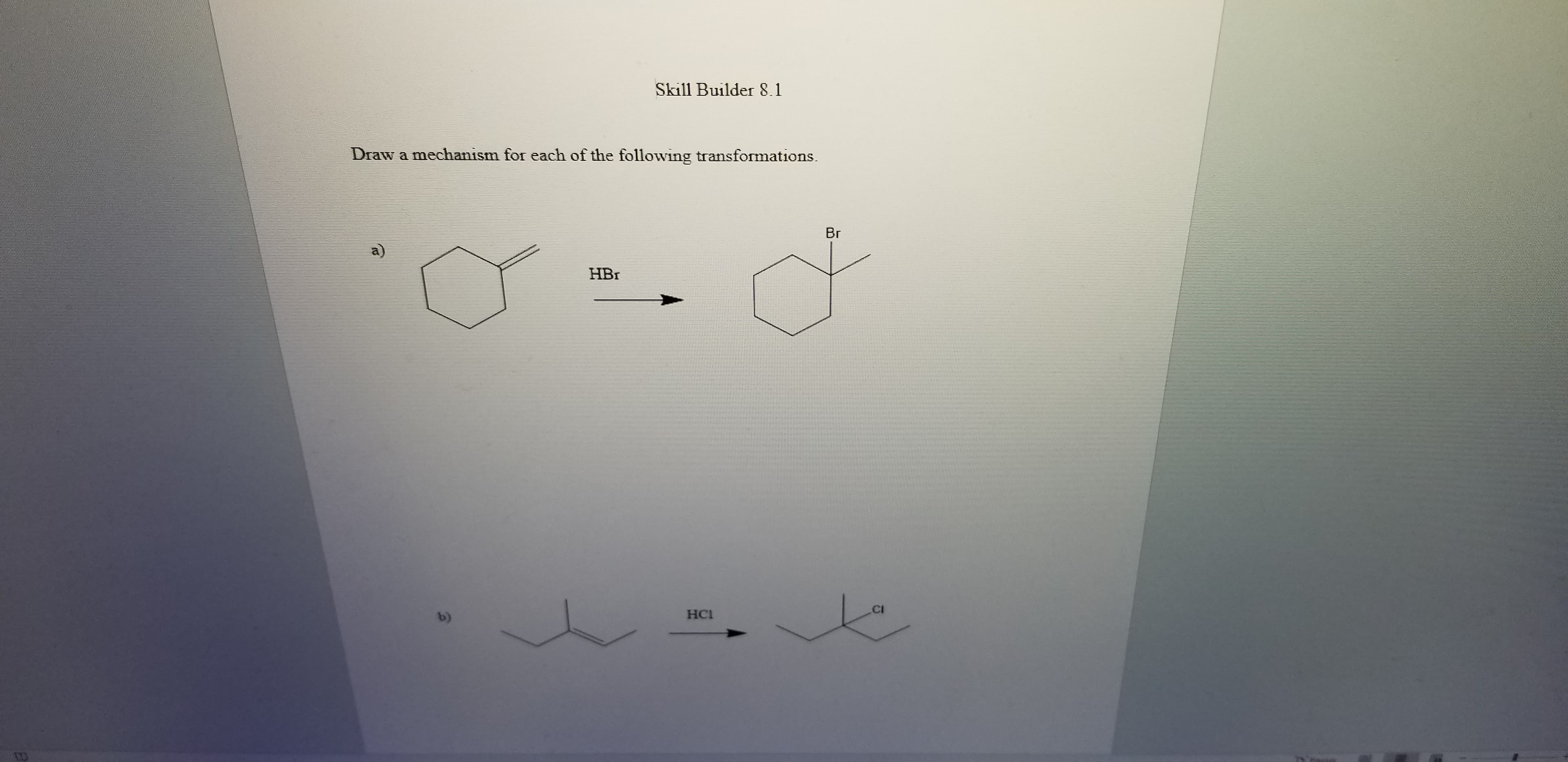
Q: (a) Br
A: Answer of this question:- In first step use anti Markovnikoff's rule . According to this rule…
Q: OH Br CH3 CH3 CH CH3 aq. H2SO4. Br2 -or- CH3 CH3 H20 option #1 option #2
A:
Q: e sure to answer all parts. raw a stepwise mechanism for the following reaction: CI CI H- HCI (2…
A:
Q: Match each reaction sequence to a product below. Assume any necessary workup. Answers may be…
A: Since you have posted multiple unrelated reaction questions that too with multiple steps, hence we…
Q: Q3: Identify reagents that can be used to accomplish each of the transformations shown below 9 7 1 2…
A: The organic reactions are carried out when the reactants react in the presence of the reagents. The…
Q: HCI (1 equiv) Draw 1,2-Product + Draw 1,4 Product
A:
Q: 4. Draw a mechanism for each of the following transformations: Br Dilute HBr a) Br, Dilute HBr (b)…
A:
Q: Draw the products of each reaction using the two-part strategy from Sample Problem 10.7.
A: EXPLANATION: The reaction of alkene with Hydrogen halide takes place by Markonikov addition.When an…
Q: Be sure to answer all parts. Predict the product and draw a stepwise mechanism for the following…
A: The given reaction will go through SN1 mechanism
Q: Draw a stepwise mechanism for the following reaction: HBr HO, Br Part 1: HOBR Br H,0 HO view…
A: Alcohol reacts with a hydrogen halide to form an alkyl halide. The reaction proceeds with an SN1…
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HBr HO. Br Part…
A: Given reaction is the reaction of alcohol with strong acid to form alkyl halide. In this reaction,…
Q: 12. Draw only the SN1 mechanism and ALL possible products for the following reaction: ar 13. Draw…
A: ->In SN1 reaction first of all carbocation formed then nucleophile added and form pair of…
Q: Draw a stepwise mechanism for the following reaction.
A: A stepwise mechanism for the following reaction is given below :
Q: Draw the product of each reaction. NH3 (excess) -NH2 CH3CH,Br (excess) a. b.
A: Draw the product of each reaction, (a) (b)
Q: -OCH3 OCH3 OH Step 1 Step 2 Step3 1 2 3
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: H* -H,0
A:
Q: 1. Consider the dehydration of the following alcohols using a Stre a circle around the alcohols that…
A: Dehydration is an elimination reaction. 10 alcohols follow E2 mechanism. 20 alcohols follow both E2…
Q: 8.30 Draw the complete, detailed mechanism (including curved arrows) for each of the following…
A:
Q: Provide an appropriate alkyne starting material A and intermediate product B. Omit byproducts. The…
A: An alkyne on reaction with R2BH ,THF will form a boron compound then the compound on reaction with…
Q: Drawing an SN1 Mechanism That Involves a Rearrangement Draw a stepwise mechanism for the following…
A: A stepwise mechanism for the following reaction has to be drawn.
Q: How many double headed red arrows are used in the last step of the E1 reaction? 0 2…
A: E1 mechanism
Q: OH PBr₂ Compound 1 PH₂ Compound 3 PBr3 ? Br Select the major product for the reaction shown below.…
A:
Q: Identify the mechanistic steps for the Claisen condensation of ethyl propionate, then draw the final…
A: In Claisen condensation reaction, a strong base abstracts a proton from alpha carbon of carbonyl…
Q: Which reagent in each of the following pairs is more nucleophilic? a) (CH3)3B and (CH3)3N b) H2O…
A: The reagent has a negative charge or lone pair of electrons acts as a nucleophile. As the…
Q: Draw the major organic product of the reaction shown. Select Draw Rings More Erase Br HBr H3C-OH
A: Alcohols primarily undergo nucleophilic substitution reactions to produce alkyl halides. This…
Q: Identify the missing reagent for the reaction below. NH2 N' CH3 NH2 `N CH3 A) ОН B) ОН C) D) CH3CH
A: Diacetyl is the reagent that exists in yellow or green color liquid. It naturally exists in…
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HBr Br Part 1:…
A: The given reaction is an example of the reaction of secondary alcohol with HBr. Generally, alcohol…
Q: C. Draw a mechanism for each of the following transformations: OH H,0+ (а) OH [H*] (c)
A: Interpretation - To draw the mechanism of each transformations which is given in the question .…
Q: 1. Show the mechanism and the direction of arrows. Label the carbons with no. if needed. A A
A: This is the Pericyclic reaction. In first step con rotation ring opening occur and second step…
Q: Draw the most stable form of the major product in the following reaction. 1. NaOC,H, 2. Н,о* С, Н,ОН
A: Given, to draw the most stable form of the major product for the following reaction :
Q: Draw the major product formed when HBr reacts with the epoxide. Use wedge-dash bonds, including…
A:
Q: Q#7 Draw a stepwise, detailed mechanism for the following reaction. Use curved arrows to show the…
A: Mechanism = To be determined
Q: 1-bromobutane and Na* OH (SN2 mechanism) 2. 3-chloropentane and K* OC(CH3)3 (E2 mechanism) 3.…
A: The reactions given are,
Q: .Which alkyl halide out of the following may follow both SN1 and SN2 mechanism? СН3-X (CH3)3C-X…
A: To identify: Which alkyl halide will react from both SN1 and SN2 mechanism.
Q: 3. Draw a mechanism to show the arrow pushing for each of the transformations shown below. NO: NO,…
A:
Q: Add curved arrow(s) to draw step 3 of the mechanism. Modify the given drawing of the product as…
A: Balance of oxygen is two,but if it's bonded with 3-atoms then it will hav ethe positive charge
Q: Br H20 Br
A: As Br is more electronegative than carbon, Br gains partial negative charge and carbon gains a…
Q: Draw the major product of the following reaction: AICI 3
A: -> Benzene can give electrophilic aromatic substitution reaction. In which hydrogen is replaced…
Q: What reagents do you need to use to for the following reactions? Match the letter with the reagent!…
A: Given conversions are epoxide ring formation from alkene and epoxide ring opening by using…
Q: 20. (10 points) Predict the product and draw the curved arrow mechanism for the transformation…
A: In the given question we have to solve the epoxide ring opening in the presence of LiAlH4 and water,…
Q: 4. Draw the mechanism of A and B reactions below using appropriate reagents. OH HO multisteps…
A: We have given that Cyclopentyl methanol react with various reagent to give product via Mechanism A…
Q: 10) Draw 1, 2, 3, and 4. 1. 4 2. 11) What reagents should be used for the reaction shown below? H. H…
A: NaNH2 is base , hydrolysis of alkyne give carbonyl compound.
Q: Q3: Identify reagents that can be used to accomplish each of the transformations shown below 8 7 1 2…
A: Reducing agents The reagents that adds Hydrogen into it is called reducing agents. Eg : 1) H2,Pd/C…
Q: 1. Propose a synthetic route from the starting material to the product. Show each individual…
A:
Q: 13 Draw a step-by-step mechanism for the transformation shown below (no additional reagents are…
A:
Q: Br NaN3 DMF Using curved arrows, draw the mechanism for the SN2 reaction below 8:55 /
A: So we’ll draw the mechanism for above given reaction. The reaction follows SN2 pathway. As we know…
Q: Draw a stepwise, detailed mechanism for the following reaction.
A: The CH3NH2 attacks on the Cl and removes it in step 1. And then in step 2, due to presence of…
Q: Be sure to answer all parts. Draw a stepwise mechanism for the following reaction: HBr Br ОН OH Part…
A:
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

- Below is a schematic representation of possible reaction that compound X can undergo.Use the scheme to answer the following questions. A.What is the IUPAC name for compound X B. What type of reactions is/are represented by (i) and(ii). C.Compound X undergo transitions through either (A) or (B) to produce compounds (1),(2),(3) and (4). Draw the structure of (A) and (B).Arrange the following compounds in order of increasing reactivity to nucleophilic addition.I. Pentan-2-one II. Pentanal III. 3-methybutan-2-one A. III, II, I B. I, II, III C. II, I, III D. III, I, IIExplain briefly by illustrations the chemistry behind each answer 1. Preamble :A reaction flask contains a 2-bromopentane in an ethanolic solution of sodium ethoxide at room temperature and results in the formation of two olefinic products(1-pentene and 2-pentene) I) What reaction pathway is followed by the reaction above?A. E2 dehydrohalogenation B. E1 dehydrohalogenationC. SN1 dehydrohalogenationD. SN2 dehydrohalogenationE. A mixture of E1 and E2 pathways II) What is responsible for the formation of different products (major and minor).A. The different activated complex involved in the mechanism.B.Bimolecular Nucleophilic substitution reaction C.Bimolecular Elimination reaction D.The presence of sodium ethoxideE.The hybridisation nature of the secondary carbocation III) All the following is true about the reaction in question 1 except?A. The reaction follows Zaistev’s ruleB. Sodium ethoxide is the nucleophile in this reactionC. The more highly substituted alkene is the most stable…
- Provide the suitable reagents to effect the following transformations. I specifically need help on sub-parts d, e, f, g, h, and i.Identify the compound in each of the following pairs that reacts at a fasterrate in an SN1 reaction.a) Isopropyl bromide or Isobutyl bromide.b) Cyclopentyl iodide or 1-Methylcyclopentyl iodide.c) Cyclopentyl bromide or 1-Bromo-2,2-dimethylpropane.Select the best reagents for each of the following transformations. Only use each set of reagents once. I specifically need help on 7 and or 6 but most specifically in 7.
- The compound below can undergo E2 reaction mechanism upon addition of sodium chloride. TRUE OR FALSE6. List the following aryl halides from most to least reactive (in suzuki coupling): 2-bromotoluene, 3-bromotoluene, 4,-bromotoluene, 4-bromoanisole, 4-bromoacetophenone, 3-chlorotoluene, and 4-chloroacetophenone1.) 2-butanol reacts with HCl in the presence of ZnCl2 at high temperature. The final product of the reaction: a. has inversion of configuration b. is racemic c. has retention of configuration d. none of these 2.) Which of the ff. halides will most probably react via an SN1 mechanism? a. CH3Cl b. CH2=CHCH2CH2Cl c. CH3CH=CHCH2Cl d. CH2=CHCl
- 1. Organometallic reagents are characterized by a. Polar bonds b. Carbon bonded to an electropositive element c. Carbon behaving as a free carbanion d. All of these 2. What is the product of the following reaction? a. I b. II c. III d. IVDuring the further development of other prostaglandin-like molecules, it is necessary to replace the cyclopentane ring with the cyclohexene as shown in the reaction scheme below. (Deuterium (D) is a heavier isotope of hydrogen) a) Draw the most stable conformation of the starting material 8 and explain why this is the most stable conformation. b) Draw the reaction mechanism for how 9 is formed from 8 and explain why 10 and 11 are not formed if one assumes the E2 mechanism.Can you explain about the order of reactivity in an E1 elimination? And for these compounds, how to list them in increasing reactivity? 1. CH2CH3CHBrCH3 2. CH3CH2CH2CH2Br 3. (CH3)3CBr

