SmartWork Ch. 16b Homewoi 1) Write the net ionic equation that represents what happens when sodium hydroxide dissolves in wat If 0.0940 moles of sodium hydroxide were added to water, how many moles of OH would be produ Number males OH 2) Which of the following reactions would occur to a measurable extent in water? Cr(aq) + H,O{) > HCI{aq) + OH (aq) F"(aq) + H;O() + HF(aq} + OH"(aq) Br (aç) + H2O() + HBr(ag) + OH"(aq) NH,"(aq) + H2O + NH3{aq) + H30*(aq)
SmartWork Ch. 16b Homewoi 1) Write the net ionic equation that represents what happens when sodium hydroxide dissolves in wat If 0.0940 moles of sodium hydroxide were added to water, how many moles of OH would be produ Number males OH 2) Which of the following reactions would occur to a measurable extent in water? Cr(aq) + H,O{) > HCI{aq) + OH (aq) F"(aq) + H;O() + HF(aq} + OH"(aq) Br (aç) + H2O() + HBr(ag) + OH"(aq) NH,"(aq) + H2O + NH3{aq) + H30*(aq)
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter4: Chemical Reactions
Section: Chapter Questions
Problem 4.32P: 4-32 When a solution of ammonium chloride is added to a solution of lead(I1) nitrate, Pb(NO3)2, a...
Related questions
Question
100%
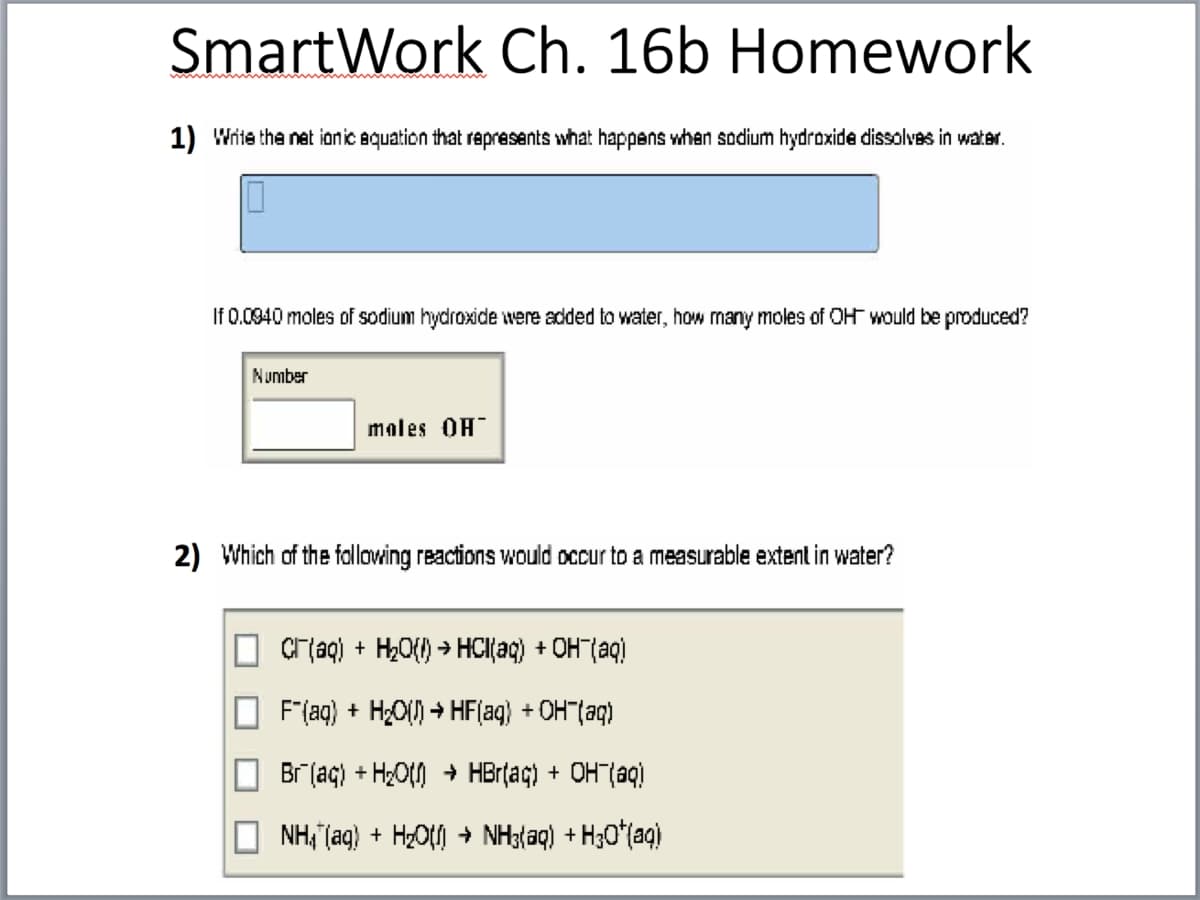
Transcribed Image Text:SmartWork Ch. 16b Homework
1) Write the net ionic equation that rapresents what happens when sodium hydroxide dissolvas in water.
If 0.0940 moles of sodium hydroxide were added to water, how many moles of OH would be produced?
Number
males OH
2) Which of the fallowing reactions would occur to a measurable extent in water?
Cr aq) + H,O{) > HCl{aq) + OH"(aq)
F"(aq) + H¿O() → HF(aq) + OH"(aq)
Br (aç) + H2Ot) + HBrțaç) + OHT(aq)
NH, (aq) + HzO + NH3(aq) + H30*(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning