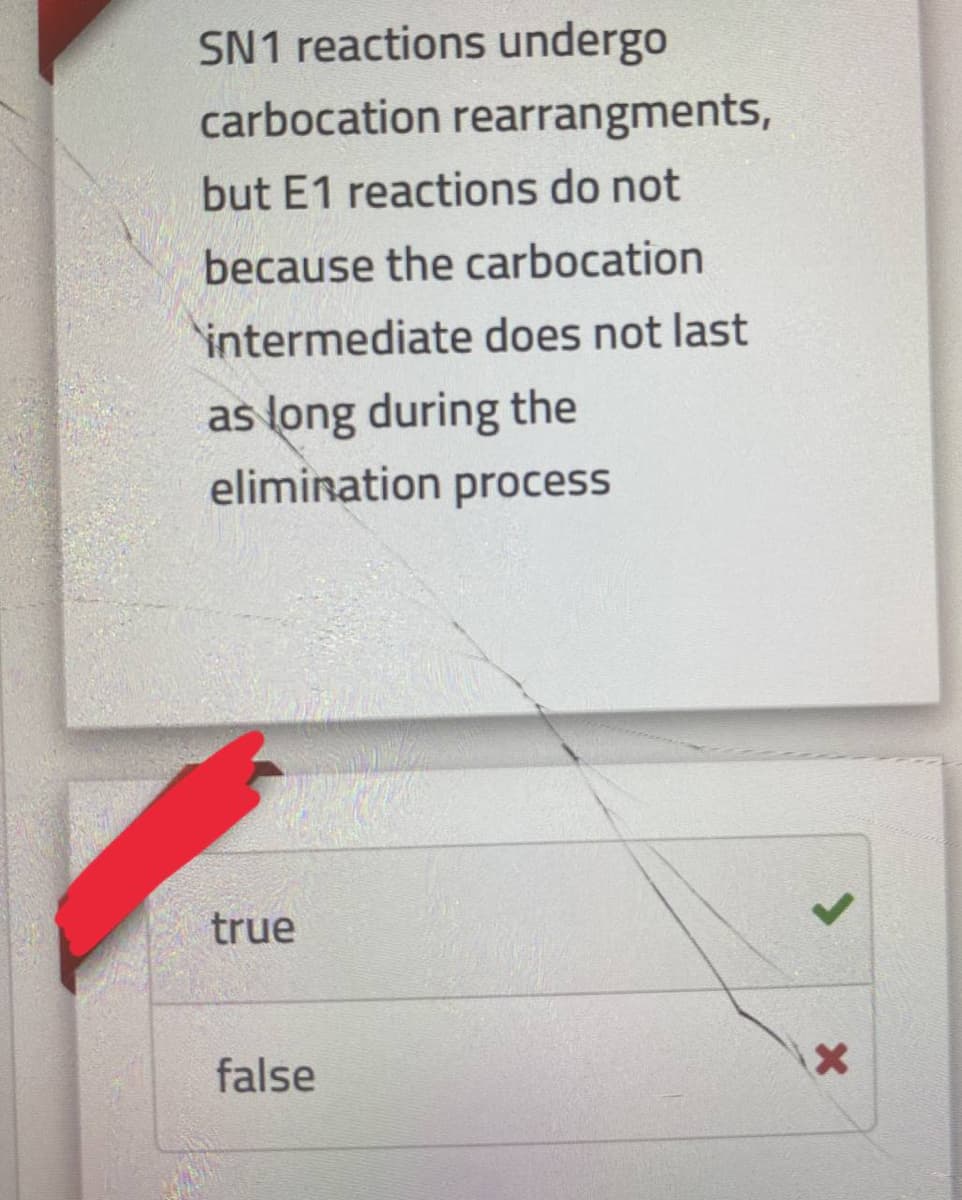
SN1 reactions undergo carbocation rearrangments, but E1 reactions do not because the carbocation intermediate does not last as long during the elimination process
Q: Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each…
A: E2 reaction is the elimination reaction bi-molecular which proceeds with anti periplanar…
Q: Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each…
A: E2 reaction is the elimination reaction in which the base can abstract the proton and…
Q: Arrange the following alky halides in order of decreasing reactivity in an SN1 reaction:…
A:
Q: Draw an alkyl halide that would undergo an SN2 reaction to yield this product under the conditions…
A: -> In SN2 Reaction nucleophile direct attack at center where leaving group present and substitute…
Q: OCH3 FeBr3 , Br2 LOCH3 Br
A: We have to draw resonance structures of anisole or the intermediate in the given reaction to…
Q: OTs Br CN Br
A: SN1 reactions are those which takes place in two steps and the first step is the rate-determining…
Q: Which of the reactions below are correctly identified? These are SN2 reactions:
A:
Q: 2. What is the major product of the following Friedel-Crafts alkylation? CH3 + CH;CHCH,CI AIC13…
A:
Q: Which undergoes faster SN1 substitution reactions with hydrogen halides, 2° or 3° alcohols? Explain…
A: Since, you have asked to solved multiple questions, so I will solved first question for you. You…
Q: Give the major elimination product for the following E2 reaction
A: E2 elimination reaction: It is a one-step reaction in which there will be a formation of an alkene…
Q: Br CH3+CH3H201H d2Br NaNz
A: Organic reaction mechanisms
Q: Predict reagents needed to complete this SN1 solvolysis reaction. Br
A: Detail mechanistic pathway for SN1 reaction is shown below
Q: Which of the following statements correctly describe(s) SN2 reactions of alkyl halides (RX)? I. Rate…
A: SN 2 reaction is nucleophilic substitution reaction.
Q: What product or products are produced in each of the following reactions? By what mechanism ( SN1,…
A: Answer:
Q: Which of the following is not a characteristic of Sn1 reactions? O the electrophilic carbon…
A: SN2 reaction proceeds through: 1. Polar protic solvent 2. Forms carbocation as an intermediate 3.…
Q: Rearrangements are likely to occur in which of the following reaction types? O E1 reactions O Both…
A: Both SN1 and E1
Q: Which of the following is NOT a good nucleophile for SN1 reactions? 1) NaOCH3 2) CH3OH 3)…
A: SN1 reactions or unimolecular nucleophilic substitution follows in two steps where in the first step…
Q: Iodoethane does not react via Sn2 reaction with NaI in Acetone. It is a good leaving group and on…
A:
Q: Given that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each…
A: Ethoxide ion is a strong base and E2 mechanism is favored by strong, negatively charged bases. It…
Q: The reaction of cyclopentanol with HBr will most likely proceed via: a. E2 b. E1 c. SN1 d. E1cb…
A: Alcohol reacts with a hydrogen halide to form an alkyl halide. The reaction proceeds with an SN1…
Q: Which solvents favor SN1 reactions and which favor SN2 reactions?
A: A chemical reaction mechanism represents the way to form and break the chemical bonds between…
Q: Which of the following can undergo E2 Elimination? Br. Br. Br Br В B&D A & C
A: Elimination reaction takes place when two atoms are removed from one molecule . At first H atom…
Q: rank the following compounds in order of their expected reactivity toward SN2 reaction CH3Br…
A:
Q: OCH3 + CH3B H;CO OCH3 H;CO OCH3 ČH3
A: Welcome to bartleby!
Q: The following reaction proceeds by two consecutive E2 eliminations. KOH, 170 °C CICH:- ethylene…
A: First, the given compound goes elimination to give an alkene which further undergoes elimination to…
Q: Which of the reactants in the picture will undergo the fastest SN1 reaction in ethanol? O D
A: Reactivity order for Alkyl halides for SN1 reaction in polar protic solvent is: Tertiary alkyl…
Q: Which of the following reactions often competes with the SN2 reaction needed to make an ylide from…
A: The correct option is (b) i.e. E2 mechanism.
Q: 3-bromo-1-pentene and 1-bromo-2-pentene undergo SN1 reaction at almost the same rate, but one is a…
A: Generally, alkyl halide or alcohols undergo elimination reaction which give rise to alkene.…
Q: Which of the statements below are correct about characteristics of an E2 reaction? i) Forms alkene…
A: Answer: E2 reaction means elimination reaction in which molecularity of rate determining step is 2.
Q: (i) Write both reactions using bond-line presentation and, using arrows providing a mechanistic…
A: Sn2 reaction is bimolecular substitution reaction.
Q: 1-Bromobicyclo[2.2.2]octane does not undergo an E2 reaction when treated with a strong base. Explain…
A: The structure of 1-bromobicyclo[2.2.2]octane is as follows:
Q: Which of the following compounds will undergo an SN1 reaction most H3)3CCH21 B) (CH3)3CC1 C)…
A: SN1 reaction are go via carbocation intermediate. More stable the formation of carbocation, faster…
Q: The elimination reaction of butan-2-ol will produce butene and but-2-ene via mechanism O a. SN1 O b.…
A:
Q: Which synthetic strategy would efficiently bring about the following transformation? ? HO- OMe OMe…
A: The following conversion take place in presence of Hg(OAc)2,H2O / NaBH4.It is oxymercuration…
Q: Which Sn2 reaction would be expected to occur most slowly? O A. CH3CH2B + CN1-i B. CH3CH21 + CN1-i…
A: More good leaving groups more will be the reactivity of alkyl halide in SN2 since order of leaving…
Q: b. Enolate chemistry facilitate many C-C bond-forming reactions, including Aldol reactions, Claisen…
A: Michael addition
Q: These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O,…
A: 1) BH3, THF, H2O2 - It is hydroboration oxidation reaction in which terminal alkyne reacted with BH3…
Q: What set of reagents can best accomplish the following transformation? A) PCC in CH2C2 B) AICI3 C)…
A: We have to tell reagent used to bring the above transformation.
Q: Rank the alkyl halides in the following marine natural product in order of increasing reactivity in…
A: Applying concept of SN1 reaction on reactivity of alkyl halide.
Q: Predict the major product in each of the following reactions and show if it originated from an SN1,…
A: Alkyl halides undergo substitution reactions absence of base, where as in presence of strong base…
Q: Define the Stereochemistry of the E2 Reaction ?
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In…
Q: Can primary alkyl halides react by SN2, SN1, E2, and E1 mechanisms? Are any of these mechanisms…
A: SN2 reaction is second order nucleophilic substitution reaction that occurs in one step and SN1…
Q: Give the major E2 product for the following reactions
A: Reaction a: In E2 eliminations more substituted olefin is major product (Zaitsev product). Reaction…
Q: For each of the following reactions indicate whether the major products will be the result of an…
A:
Q: 2. Explain why the following deuterated 1-bromo-2-methylcyclohexane undergoes dehydrohalogenation by…
A:
Q: Br CH3OH +
A: Both are SN1 reactions because both the substrates are able to generate the tertiary…
Q: Rank the following compounds in order of decreasing SN1 reactivity?
A: carbocation intermediate stability order : 3°>2°>1°
Q: 2) Rank these molecules by their SN2 reaction rate. If a difference in reaction rate is hard to…
A: The correct order of molecules towards SN2 reaction is D > E > A > C > B. The order…
Q: Rank these substrates in order of increasing ability to undergo a nucleophilic substitution aromatic…
A:
Step by step
Solved in 2 steps

- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.) Propose a mechanism to account for formation of both A and B1. SN1 or SN2 2. Include major product and stereochemistry 3. Add detailed reaction mechanismsFor each of the following reactions give the Major product(s) of the reaction only and statethe mechanism the reaction follows (SN1, SN2, E1, E2).
- Tertiary alkyl halides will react by E2 under neutral conditions under acidic conditions under basic conditions will not react by E2Explain how and why rearrangements occurduring Friedel-Crafts alkylation reactions formingmore than 1 product. Also illustrate therearrangement reaction from the aboveexample.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below
- The following compound undergoes Benzilic Acid Rearrangementto yield ahydroxyacid salt. Proposea mechanism for the reaction, writethe major product,and provide an explanation as tothe preference of migration of one R group over the other.Complete the following reactions, write major product(s) and specify the mechanism if; SN1 SN2, E1, or E2The reaction H2C=CHCH2Br + NaN3 followed by reaction with LiAlH4 yields CH3CH2CH2NH2 H2C=CHCH2NH2 H2C=CHCH2N3 none of the above
- What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?Draw Sn1/E1 products of both reactions below or fill in the blank with appropriate stuffs for Sn1/E1Competing SN and E reactions. Predict whether the following reactions will proceed via substitution (SN1 or SN2), or elimination (E₁ or E₂), or whether the two will compete. Identify the major product.

