Chapter10: Organohalides
Section10.2: Preparing Alkyl Halides From Alkanes: Radical Halogenation
Problem 4P: Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s)...
Related questions
Question
Which of the reactions below are correctly identified?
|
These are SN2 reactions:
|
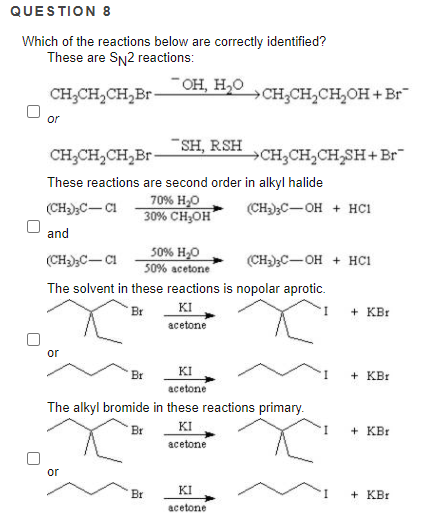
Transcribed Image Text:QUESTION 8
Which of the reactions below are correctly identified?
These are SN2 reactions:
- он, н,о
CH;CH,CH,Br-
CH;CH,CH,OH + Br
or
SH, RSH
CH;CH,CH,Br-
CH;CH,CH,SH+Br"
These reactions are second order in alkyl halide
70% H,0
30% CH;OH
(CH);C- CI
(CH-);C-OH + HC1
and
50% H,O
50% acetone
(CH)C-CI
(CH3)3C-OH + HC1
The solvent in these reactions is nopolar aprotic.
KI
acetone
Br
+ KBr
or
Br
KI
+ KBr
acetone
The alkyl bromide in these reactions primary.
Br
KI
+ KBr
аcetone
or
Br
KI
+ KBr
acetone
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



