Solubility Exceptions soluble Multiple Choice: lon NO3- none CIO4- soluble soluble soluble none except Ag+, Hg22+, *Pb2+ except Ag+, Hg22+, *Pb2+ except Ag+, Hg22*, *Pb2+ except Ca2+, Ba2+, Sr2+, Hg2+, Pb2+, CI- Br- SO,2- soluble 1. Which of the following pairs of ions, when mixed, would form a precipitate? soluble Ag+ CO32- insoluble except Group IA and NH4+ PO43- insoluble except Group IA and NH4 insoluble except Group IA, *Ca²+, Ba2+, Sr2+ insoluble except Group IA, IIA and NH4+ a. Cu2+, I- OH- b. Ba2+, S2- S2- Nat soluble None c. Ca2+, OH- d. Sn2+, CH K+ soluble None NH4+ soluble None 2. The balanced equation that show iron (II) sulfate dissolved in water is a) Fe2(SO4)3(s) → 2Fe3+(aq) + 3SO42(aq) b) Fe2(SO4)3(s) → Fe2(aq) + (SO4)3(aq) c) Fe2(SO4)3(s) →2F22*(aq) + 3SO,3 (aq) d) Fe2(SO4)3(s) + H20(1) → H2SO4(aq) + Fe(OH)3(aq) 3. How many moles are in a 25.0mL sample of a 0.10OM solution? a. 0.00250moles C. 2.50 moles b. 1.00moles d. 4.00 moles
Solubility Exceptions soluble Multiple Choice: lon NO3- none CIO4- soluble soluble soluble none except Ag+, Hg22+, *Pb2+ except Ag+, Hg22+, *Pb2+ except Ag+, Hg22*, *Pb2+ except Ca2+, Ba2+, Sr2+, Hg2+, Pb2+, CI- Br- SO,2- soluble 1. Which of the following pairs of ions, when mixed, would form a precipitate? soluble Ag+ CO32- insoluble except Group IA and NH4+ PO43- insoluble except Group IA and NH4 insoluble except Group IA, *Ca²+, Ba2+, Sr2+ insoluble except Group IA, IIA and NH4+ a. Cu2+, I- OH- b. Ba2+, S2- S2- Nat soluble None c. Ca2+, OH- d. Sn2+, CH K+ soluble None NH4+ soluble None 2. The balanced equation that show iron (II) sulfate dissolved in water is a) Fe2(SO4)3(s) → 2Fe3+(aq) + 3SO42(aq) b) Fe2(SO4)3(s) → Fe2(aq) + (SO4)3(aq) c) Fe2(SO4)3(s) →2F22*(aq) + 3SO,3 (aq) d) Fe2(SO4)3(s) + H20(1) → H2SO4(aq) + Fe(OH)3(aq) 3. How many moles are in a 25.0mL sample of a 0.10OM solution? a. 0.00250moles C. 2.50 moles b. 1.00moles d. 4.00 moles
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.18QAP
Related questions
Question
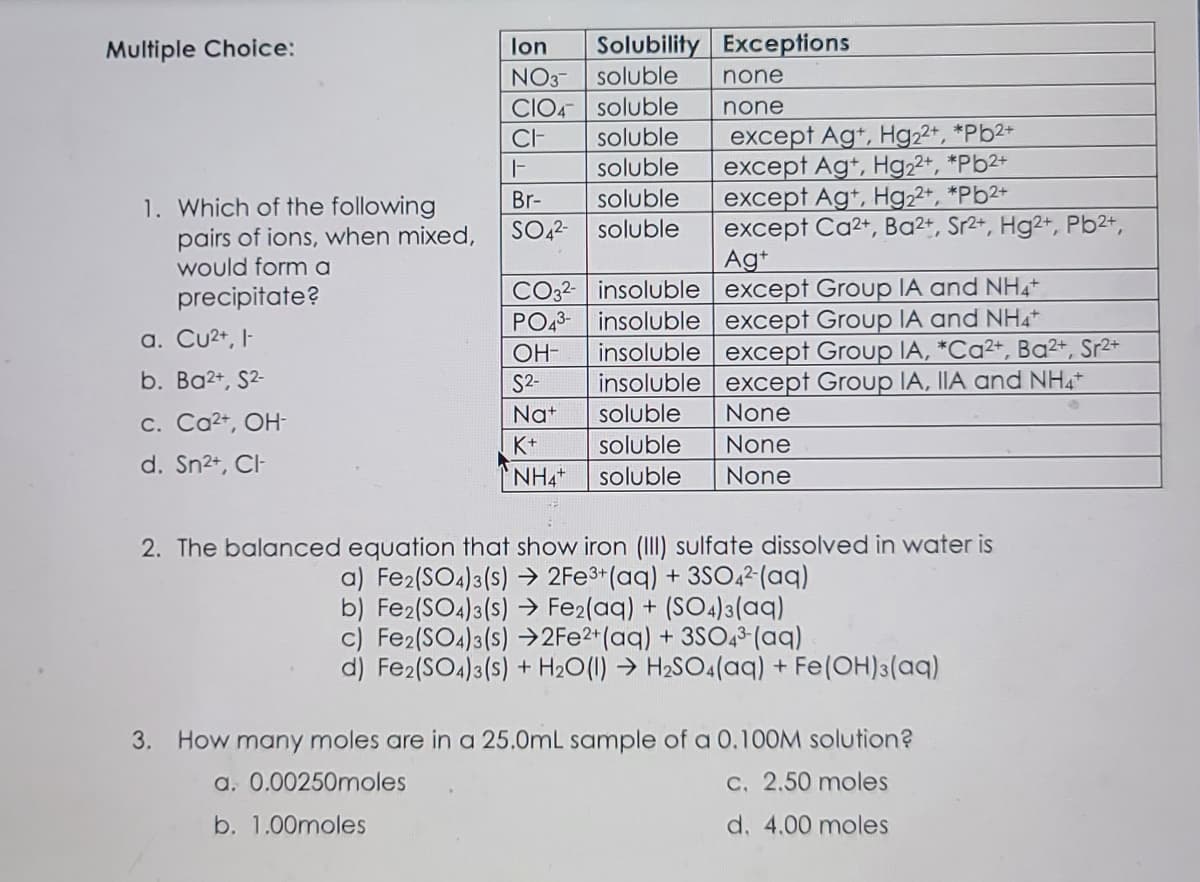
Transcribed Image Text:Multiple Choice:
lon
Solubility Exceptions
NO3-
soluble
none
CIO4- soluble
soluble
soluble
none
except Agt, Hg22+, *Pb2+
except Ag+, Hg22+, *Pb2+
except Ag+, Hg22*, *Pb2+
except Ca2+, Ba²+, Sr2+, Hg2+, Pb2+,
CI-
Br-
SO,2-
soluble
1. Which of the following
pairs of ions, when mixed,
would form a
precipitate?
soluble
Ag+
CO32- insoluble except Group IA and NH4
insoluble except Group IA and NH4
insoluble except Group IA, *Ca²+, Ba²+, Sr2+
insoluble except Group IA, IIA and NH4+
PO43-
a. Cu2+, I-
OH-
b. Ba2+, S2-
S2-
Nat
soluble
None
c. Ca2+, OH-
K+
soluble
None
d. Sn2+, CI
NH4
soluble
None
2. The balanced equation that show iron (III) sulfate dissolved in water is
a) Fe2(SO4)3(s) → 2FE3*(aq) + 3SO,²(aq)
b) Fe2(SO4)3(s) → Fe2(aq) + (SO4)3(aq)
c) Fe2(SO4)3(s) →2F22*(aq) + 3SO,3 (aq)
d) Fe2(SO4)3(s) + H20(1) → H2SO4(aq) + Fe(OH)3(aq)
3. How many moles are in a 25.0mL sample of a 0,10OM solution?
a. 0.00250moles
C. 2.50 moles
b. 1.00moles
d. 4.00 moles
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

