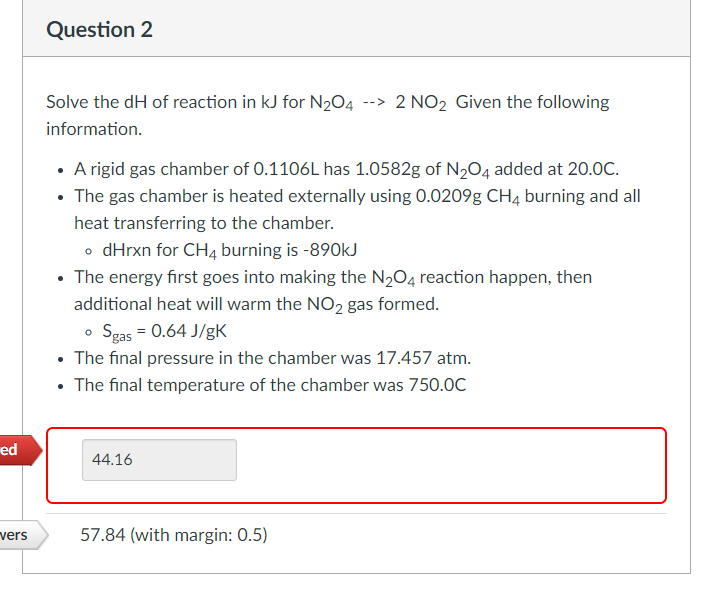
Solve the dH of reaction in kJ for N₂O4 --> 2 NO2 Given the following information. • A rigid gas chamber of 0.1106L has 1.0582g of N₂O4 added at 20.0C. • The gas chamber is heated externally using 0.0209g CH4 burning and all heat transferring to the chamber. • dHrxn for CH4 burning is -890kJ • The energy first goes into making the N₂O4 reaction happen, then additional heat will warm the NO₂ gas formed. • Sgas = 0.64 J/gK • The final pressure in the chamber was 17.457 atm. The final temperature of the chamber was 750.0C
Solve the dH of reaction in kJ for N₂O4 --> 2 NO2 Given the following information. • A rigid gas chamber of 0.1106L has 1.0582g of N₂O4 added at 20.0C. • The gas chamber is heated externally using 0.0209g CH4 burning and all heat transferring to the chamber. • dHrxn for CH4 burning is -890kJ • The energy first goes into making the N₂O4 reaction happen, then additional heat will warm the NO₂ gas formed. • Sgas = 0.64 J/gK • The final pressure in the chamber was 17.457 atm. The final temperature of the chamber was 750.0C
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter5: Principles Of Chemical Reactivity: Energy And Chemical Reactions
Section5.8: Product- Or Reactant-favored Reactions And Thermodynamics
Problem 1.2ACP
Related questions
Question
Transcribed Image Text:ed
vers
Question 2
Solve the dH of reaction in kJ for N₂O4 --> 2 NO₂ Given the following
information.
• A rigid gas chamber of 0.1106L has 1.0582g of N₂O4 added at 20.0C.
• The gas chamber is heated externally using 0.0209g CH4 burning and all
heat transferring to the chamber.
• dHrxn for CH4 burning is -890kJ
• The energy first goes into making the N₂O4 reaction happen, then
additional heat will warm the NO2 gas formed.
o Sgas = 0.64 J/gK
• The final pressure in the chamber was 17.457 atm.
The final temperature of the chamber was 750.0C
44.16
57.84 (with margin: 0.5)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning