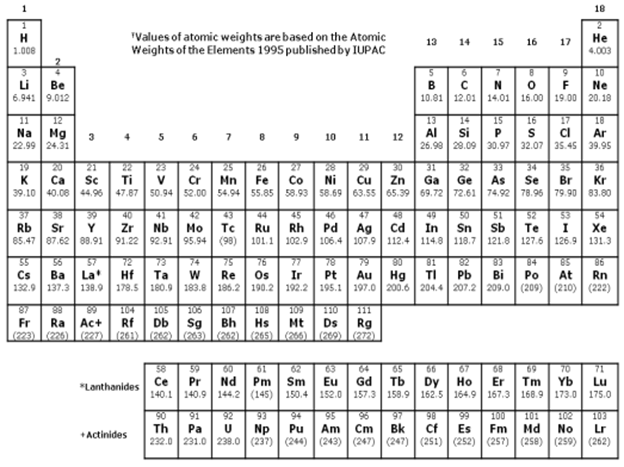
Solve the following problems and show complete solutions. The final answers must be reported in proper units and number of significant figures. Assume that all solutions were prepared at 25°C, and the solutes were completely dissolved. Molar masses should be based on the Periodic Table provided. Density values are provided if needed. Alcoholic beverages are solutions. Typically, a beer contains about 4.50% (v/v) of ethanol fermented by yeast from sugars, while a particular soju brand contains about 24.0% (v/v) ethanol. A Korean-inspired drink was prepared by mixing 500.0 mL of beer with 200.0 mL of the soju. The mixture was finally diluted to 750.0 ml using enough cola. a. Compute the final concentration (%w/v) of ethanol in the drink. b. How much of the drink (in mL) must a person take all at once so that the amount of ethanol ingested is 25.0 mL? DENSITY VALUES AT 25°C Ethanol: 0.789 g/mL Acetic Acid: 1.05 g/mL Water: 0.997 g/mL
Solve the following problems and show complete solutions. The final answers must be reported in proper units and number of significant figures. Assume that all solutions were prepared at 25°C, and the solutes were completely dissolved. Molar masses should be based on the Periodic Table provided. Density values are provided if needed. Alcoholic beverages are solutions. Typically, a beer contains about 4.50% (v/v) of ethanol fermented by yeast from sugars, while a particular soju brand contains about 24.0% (v/v) ethanol. A Korean-inspired drink was prepared by mixing 500.0 mL of beer with 200.0 mL of the soju. The mixture was finally diluted to 750.0 ml using enough cola. a. Compute the final concentration (%w/v) of ethanol in the drink. b. How much of the drink (in mL) must a person take all at once so that the amount of ethanol ingested is 25.0 mL? DENSITY VALUES AT 25°C Ethanol: 0.789 g/mL Acetic Acid: 1.05 g/mL Water: 0.997 g/mL
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.1E
Related questions
Question
4.
Kindly solve for me to understand.
Transcribed Image Text:18
2
"Values of atomic weights are based on the Atomic
Weights of the Elements 1995 published by IUPAC
Не
13
14
15
16
17
1.008
4.003
10
Li
Be
6.941 9.012
B
10.81 12.01
F
Ne
19.00 20.18
14.01
16.00
11
12
13
14
15
16
17
18
Na Mg
3
6.
10
11
12
Al
Si
ci
Ar
22.99 24.31
26.98 28.09 30.97 32.07 35.45 39.95
22
23
24
25
Mn
27
28
29
30
20
Са
39.10 40.06 44.96
TE
32
Ge
EE
As
ÞE
35
Br
19
21
26
36
Sc
Ti
47.87 50.94 52.00 54.94 55.85 58.93 58.69 63.55 65.39 69.72 72.61 74.92 78.96 79.90 83.80
V
Čr
Fe
Co
Ni
Cu
Zn
Ga
Se
Kr
37
38
39
40
41
42
43
44
45
46
47
48
49
50
51
52
$3
54
Zr
85.47 87.62 88.91 91.22 92.91 95.94
Ru
To
(98)
101.1 102.9 106.4 107.9 112.4 114.8 118.7 121.8 127.6 126.9 131.3
Xe
Rb
Sr
Nb
Mo
Rh
Pd
Ag
Cd
In
Sn
Sb
Te
78
81 2 83
Pb
65
At
55
56
57
72
Hf
178.5 180.9 183.8 186.2 190.2| 192.2 195.1
73
Ta
74
75
76
77
79
80
84
86
Cs
Ва
La
Re
Os
Ir
Pt
Au
197.0 200.6 204.4 207.2 209.0 (209) (210) | (222)
Hg
TI
Bị
Po
Rn
132.9 137.3 138.9
97
Fr
228) (226) (227) (261) (262) (263) (262) (265) (266) (260) (272)
104
105
Db
106
107
108
109
Mt
110
111
Ra
Ac+
Rf
Sg
Bh
Hs
Ds
Rg
64
Gd
140.1 140.9 144.2 (145) 150.4 152.0 157.3 158.9 162.5 164.9 167.3 168.9 173.0 175.0
59
Pr
60
Nd
61
Pm
62
Sm
63
Eu
65
Tb
67
68
Er
69
Tm
70
58
Ce
71
Lu
66
*Lanthanides
Dy Ho
Yb
97
Bk
232.0 231.0 238.0 (237) | (244) (243) (247) (247) (251) (252) (257) (258) (259) (262)
91
92
93
94
Pu
95
Am
98
Th Pa
99
Es
100
Fm
101
Md
102
No
103
Lr
Np
Cm
cf
*Actinides
9.

Transcribed Image Text:Solve the following problems and show complete solutions. The final answers must be reported in
proper units and number of significant figures. Assume that all solutions were prepared at 25°C, and
the solutes were completely dissolved. Molar masses should be based on the Periodic Table provided.
Density values are provided if needed.
Alcoholic beverages are solutions. Typically, a beer contains about 4.50% (v/v) of ethanol
fermented by yeast from sugars, while a particular soju brand contains about 24.0% (v/v)
ethanol.
A Korean-inspired drink was prepared by mixing 500.0 mL of beer with 200.0 mL of the soju. The
mixture was finally diluted to 750.0 mL using enough cola.
a. Compute the final concentration (%w/v) of
ethanol in the drink.
b. How much of the drink (in mL) must a
person take all at once so that the amount of
ethanol ingested is 25.0 mL?
DENSITY VALUES AT 25°C
Ethanol: 0.789 g/mL
Acetic Acid: 1.05 g/mL
Water: 0.997 g/mL
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning