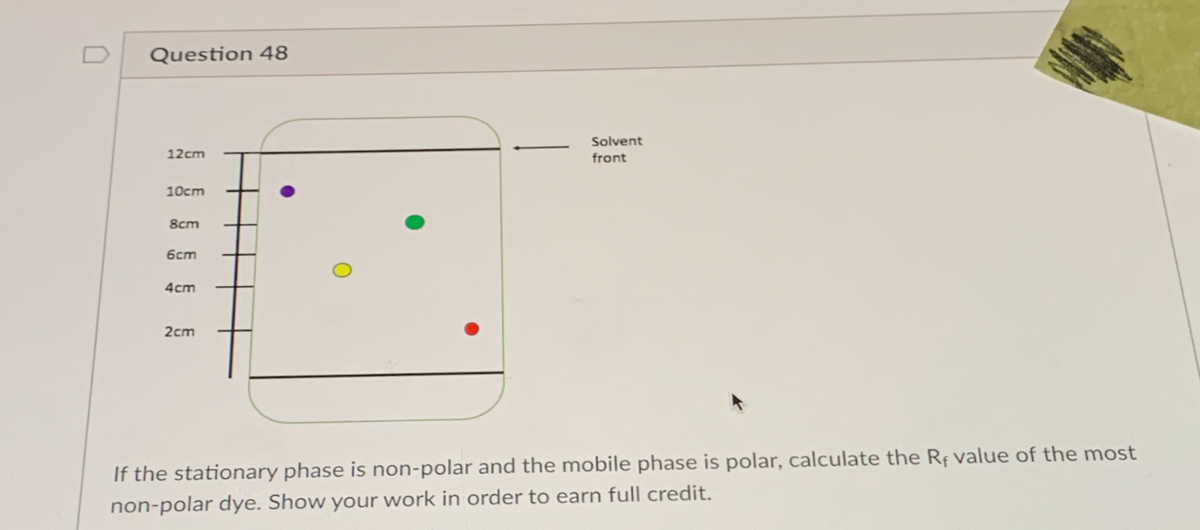
Solvent 12cm front 10cm 8cm 6cm 4cm 2cm If the stationary phase is non-polar and the mobile phase is polar, calculate the Rf value of the most
Q: I need help with this problem. Draw the rotation or reflection of the XeF4. sigma h of XeF4 and…
A:
Q: 13-when changing the polarityof solvent frome non-polar .the value of the transition wavelength…
A: In the given question we have to choose the correct option from the following. when changing the…
Q: On the Jablonski diagram below, draw and label the following interactions. a. 1 photon fluorescence…
A:
Q: © fnd the tanetion metel element (M) m) facllening luitble Valance eleeban Count [M3 (co),z] C three…
A:
Q: Which THREE Miller planar indices' labels are INCORRECT? A (11 1) в [112] c(133) 24 Z4 0.5 +Y +Y 1/3…
A: Miller indices is a notation system in crystallography for lattice planes in crystals. It is…
Q: A if the value in column l is greater than in II B if the value in column II is greater than in I…
A: As R and S does not contain any specific values so 70%R and 30%S , 70%A and 30%R have same amount of…
Q: A. Solve for the Rf value 400mm 333mm 222mm 111mm 99mm 55mm 22mm 5mm A B C D E B. Arrange the…
A: The Compound which have more polarity then it’ll travel smaller distance from baseline and more…
Q: OC -Fe: co What modes of the CO stretching of this molecule (Fel2(CO)3 with L axial) are IR active?…
A: In order to solve this question, we will find out the point group of this molecule and then identify…
Q: a polar compound will have a (smaller/larger) Rf when a polar stationary phase is used
A: Rf value is given by: Rf=distance travelled by given componentdistance travelled by solvent front
Q: 3. PPM 4. -LO
A: below answer is given chemical shift values are written from spectra
Q: Prepare a representation flowchart for trans- N2F2, which has C2h symmetry.
A: N2F2 is the chemical formula of Dinitrogen difluoride. It exists as gas state at room temperature…
Q: 6. Complete the following graph of potential energy vs. rotation of the C-C2 bond of ethane. Do not…
A: While drawing a Newman projection for a certain molecule, it is usually viewed along C-C bond. In…
Q: 3. Indicate which arrow-pushing schemes below show proper use of curved arrows and which do not.…
A: In the arrow pushing scheme of any reaction the electron rich atom attacks on the electron deficient…
Q: Which of the following arrow movements are wrongly ordered?
A: Tail to head of arrow in the mechanism of a chemical reaction shows the movement of electrons (from…
Q: Can you help me predict the major product of for the following reaction
A: The reaction is
Q: Find the correct order of stretching frequency of O-O bond in the following molecules: O2, 02(-),…
A:
Q: :O: FCH2 (а) CH2 (b) III II 0.
A: If there is conjugation system in molecule then resonance occurs.There are various structures…
Q: Which stable molecules have their highest-energy electrons in antibondingorbitals?
A: From the given, Ne2 does not exist because the bond order for this molecule is zero.
Q: In each of the following molecules 1-6, identify the relation between the circled hydrogen atom as…
A: On the basis given questions , I have find the relationship between circle Hydrogen.
Q: Which one of the following would be paramagnetic? Group of answer choices C2 B2 F2 N2
A: Concept is based on MOED(molecular orbital energy level diagram).
Q: MCQ 22: The name given to sub-shells are s-sharp, p-principal and d for A. different B. diffuse C.…
A: The given options are,
Q: Evaluate the quotient. 4.21×10^4 divided by 7.77 x 10^7
A: Given that : We have to evaluate the quotient for the following : 4.21×104 divided by 7.77 x 107
Q: . shift tab fn MISSED THIS? Read Section 11.2 (Pages 438-442), 11.3 (Pages 442-447): Watch KCV 11.2.…
A: Solution given in step 2.
Q: 1. Explain nuclear magnetic moment does not align do not immediately align with applied magnetic…
A: The nuclear magnetic moment is the moment of an atomic nucleus and arises from the spin of the…
Q: how many IR bands are there in PdL2Cl2?
A: We have to identify IR bands are there in PdL2Cl2 as follows in step 2:
Q: Calculate delta H for the graph below. y = -59.88x -1.6547 -1.84 -1.645 -1.85 1.800 Ink vs 1/T…
A: The equation for a linear graph is given by y=mx + c where m is the slope and c is the y-intercept.
Q: Mearon gire cwhy the pünt gooup 18 Czy give defail ex planation uh point dragsam for each gooup.
A: Here we have to explain above point group of compounds-
Q: Which of the structures have a major peak between 1700 and 3000 cm1? (Select all that apply). none…
A:
Q: Draw all products, including stereoisomers, in each reaction.
A: Sorting among SN2,SN1,E2 and E1 Step 1: Check nucleophile/base If neutral, then SN1/E1 ------>…
Q: tersections. a¡ = 1, az= -1/2, a3 = 1 , c = ∞ and draw it.
A:
Q: FAB ion mass detection quadrupole
A: Mass spectrometry is an analytical technique.
Q: 3D drawing for 1. NH4+ 2. PCl5 3. SeF4 4. Tel6 5. XeF4
A:
Q: Draw the molecular dipole or demonstrate the symmetrical cancellation of vectors in the following…
A: Answer : Correct option is 'F'. And CF2Cl2 is a polar molecule.
Q: An unknown compound is believed to be K2[Co(CN)4] or K3[Co(CN)6] is analyzed and found to have a…
A: Given : The two possibility of the formula of unknown compound are K2[Co(CN)4] and K3[Co(CN)6] And…
Q: 7) Consider a solution of polymer molecules of length N. Find an expression for the overlap…
A: c* denotes the critical overlap concentration of polymer coils, which is an important…
Q: paramcgnetic: mdek
A: (a)An isolated gas phase Cr3+ ion Cr atomic number =24electronic configuration =[Ar]3d54s1 Cr3+…
Q: Consider the following eqm: SO2(g) + NO2(g) ⇌ NO(g) + SO3(g) What effect will adding SO3 have on the…
A:
Q: 2 Balanie! the sollowing equatios Hzi + -> HzO 2.1 H2Oz + HzO 2.2 so3 2.3. sOz ->
A: Following are the balanced Chemical equation of the given reactions.
Q: What is the F-Xe-F bond angle in XeF4? Show work
A: XeF4 has a square-planar geometry.
Q: Write a molecule and draw its shape with Cs point group
A:
Q: Which of the structures have a major peak above 3000 cm1? (Select all that apply). none of these
A:
Q: 8. Match the molecule with the correct combination of Amdextinction coefficient. A. 194 nm (s =…
A:
Q: The LiH dipole torque is 1.964 * 〖10〗^(-30) Cm and the bond length is 1.596 A. Find the percentage…
A: Given Dipole Torque = 1.964 * 10-30 CmBond length = 1.596 A° = 1.596 * 10-10 m
Q: STANDARD REDUCION POINTIALS AT25C NaYage Na) -2.713 -2.356 -1676 -0.763 Zaag)+2e Zn() Fe"(ag)2e-Fel)…
A: Reducing agents are the agents which can undergo oxidation and reduces the other compound or…
Q: Draw the molecular dipole or demonstrate the symmetrical cancellation of vectors in the following…
A: The molecule will be polar if the net dipole moment is not zero. The molecule will be non-polar if…
Q: if peter wants to remove the effect of the matrix because of the interference it causes, he should…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: 1 HS 2. SH
A: The IUPAC names of given compounds has to be given.
Q: "СООН v Pi delocalization A. Present CH hyperconjugation B. Absent Lone-pair delocalization…
A:
Q: Tiags to resolve the QC problem.
A:
Q: CH3 CH3 CH3 H3C. CH3
A: Simple definition of strain is an increase in molecular potential energy due to electron repulsion.…
Step by step
Solved in 2 steps

- 50 uL of an aqueous sample of double-stranded DNA is dissolved in 950 uL of water. This diluted solution has a maximal absorbance of 0.326 at 260 nm. What is the concentration of the original (more concentrated) DNA sample, expressed in ug/uL?True or False 1. Sample preparation of infrared spectroscopy uses KBr as the blank because Kbr has 100% transmittance at the IR wavelength range. 2. The electronegativity difference present in a dipole moment within a bond is directly proportional to the electromagnetic field produced. 3. The electromagnetic radiation that is emitted in infrared spectroscopy is not enough to excite electrons to an unoccupied molecular orbital.Unrounded Rounded ε∗,L/μmol 0.0259825 0.0260 Heres the data if needed: TZ # Concentration Absorbance at 430 nmnm 1 33.6480 0.931 2 25.2360 0.757 3 16.8420 0.210 4 8.41200 0.137 5 4.20600 0.122
- Q. based on the graph, the absorbance of caffeine using UV-vis spectroscopy is at ________. CHOICES: A. 320 nm B. 260 nm C. 260-290 nm D. 275 nm E. 260-300 nmWhich nmr active nuclei have lowest natural abandance 15N 13C 19F 31pPredict the splitting pattern for each kind of hydrogen in isopropyl propanoate, CH3CH2CO2CH(CH3)2.
- Identify the remaining three fragments from the table. (m/z=22,16,12) (hint: for m/z=22, the value of Z isn't necessarily 1)The electronic configuration of 15 X 3- ion is a. [Ne]3s23p2 b. [Ne]3s2 c. [Ne]3s23p6 d. [Ne]3s23p1Bartleem is a chiral molecule. At 70 oC Bartleem is a liquid with a density of 0.722 g/mL. At this temperature and using a 589 nm light source with a 1 dm path length the rotation of light is found to be 71o. What is the optical rotation for Bartleem?
- Q.No.2 What is spectroscopy?. What types of analysis you can do with this technique ?. Write a common procedure that you can adapt when you will perform any analysis with Spectroscopy.1. The slope of a standard curve of Abs vs [protein] is equalivent to which Lambert-Beer variable? A.) OD B.) ϵ C.) c D.) D 2.) DNA absorbs light around 260 nm. What wavelength do proteins absorb? A.) 625 nm B.) 140 nm C.) 240 nm D.) 280 nmPlease refer to the image attached below and answer the following question. Thanks What referrals would you give to her and in what order?

